Johnson & Johnson (J&J) MedTech has today announced 12-month results from the SmartfIRE study, delivered as a late-breaking presentation at the ongoing European Heart Rhythm Association (EHRA) congress (30 March–1 April 2025, Vienna, Austria), with findings showing that use of the company’s dual-energy Thermocool Smarttouch SF catheter was 86.9% effective when patients were treated with high adherence to the recommended ablation parameters. Also observed were a clinically meaningful improvement in quality of life and reduced healthcare utilisation, as well as a significant reduction in total antiarrhythmic drug (AAD) use.
The dual-energy Thermocool Smarttouch SF catheter is an irrigated, contact force-sensing catheter powered by J&J MedTech’s Trupulse generator, and is fully integrated with the company’s Carto 3 system for advanced electro-anatomical mapping and tag indexing. According to J&J MedTech, the platform is the first dual-energy pulsed field ablation (PFA) and radiofrequency (RF) ablation catheter integrated with a PFA and RF ablation index, which have proved to be important factors for durable lesion formation.
The dual-energy Thermocool Smarttouch SF catheter—which received European CE-mark approval in December 2024 and is currently investigational in the USA—has been designed on the “familiar platform” of the RF Thermocool Smarttouch SF catheter, with many years of physician experience, the company further claims.
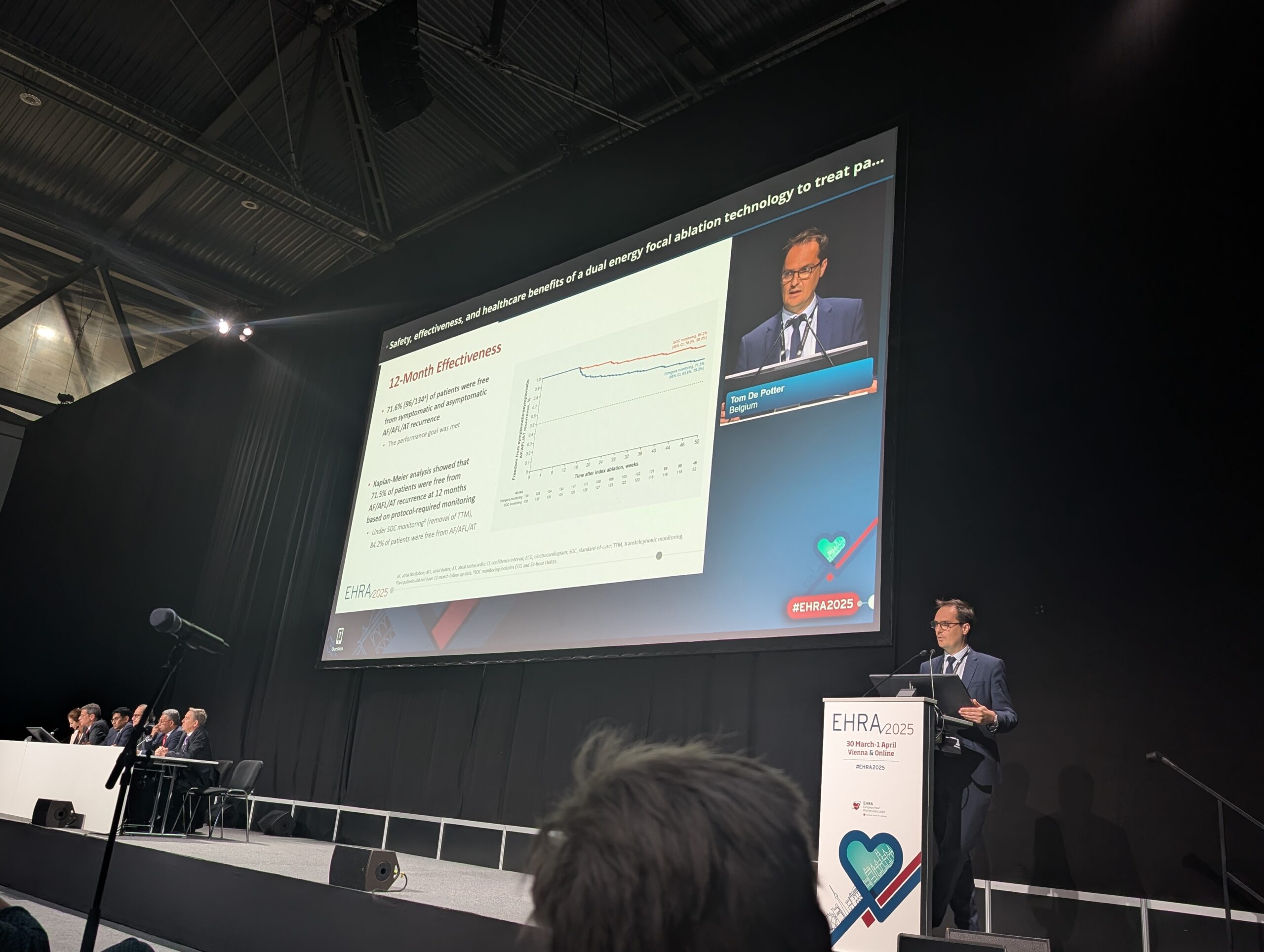
SmartfIRE was a first-in-human study assessing the safety and effectiveness of the dual-energy Thermocool Smarttouch SF catheter for the treatment of drug-refractory, symptomatic paroxysmal atrial fibrillation (AF). Primary endpoints were met at the three-month follow-up, with the latest findings being observed among eligible participants completing 12-month follow-up, including:
- A 71.5% rate of 12-month freedom from recurrence with protocol-required, stringent monitoring; this rate increased to 86.9% among patients who were treated with high adherence to the recommended ablation workflow
- A 3.6% serious adverse device effects (SADE) rate; the SADEs were not classified as related to PF energy
- A 40.8% relative reduction in cardiovascular-related hospitalisations compared to baseline
- A 60.4% relative reduction in Class I/III AAD use compared to baseline, with a significant reduction in total AAD use observed
“With the global incidence of AF rising due to an ageing population and the increasing prevalence of cardiovascular risk factors, there is a growing need for advanced, integrated technologies that support cardiac ablation procedures with clinical precision, proven efficacy, and optimised procedural efficiency,” said Tom De Potter (Onze Lieve Vrouwziekenhuis [OLV] Hospital, Aalst, Belgium), who presented these results for the first time at EHRA 2025. “With an efficacy rate of 86.9% with high adherence to the recommended ablation protocol leveraging the PF and RF tag index, the 12-month SmartfIRE data support the effectiveness, safety, and clinical benefits, of ablation performed with the dual-energy Thermocool Smarttouch SF catheter.”
J&J MedTech says it is also supporting the presentation of real-world evidence data from the company-sponsored REAL AF registry—a database comprised of 68 centres investigating real-world safety and effectiveness of catheter ablation procedures, health outcomes, research, and the future of electrophysiology practice. The data presented from the REAL AF registry include those examining gender differences in ablation strategies and outcomes, and outcomes with zero fluoroscopy versus conventional approaches.
The company is supporting additional studies with the dual-energy Thermocool Smarttouch SF catheter as well as a number of investigator-initiated collaborative studies. The first patients have been enrolled in the GPfIRE collaborative study evaluating the impact of PF energy, both alone and in combination with RF energy, on vagal modulation after pulmonary vein isolation (PVI) in paroxysmal AF. According to J&J MedTech, activation of the autonomic nervous system can induce changes to atrial electrophysiology—and GPfIRE is designed to assess how ablation with PF and RF energy affects the autonomic nervous system and, in turn, heart rate variability. The first cases in the study were also performed by De Potter at OLV Hospital.
“At Johnson & Johnson MedTech, we recognise that advancing patient care requires strong collaborations and rigorous clinical research,” commented Jennifer Currin, vice president of scientific affairs at J&J MedTech. “By supporting studies like GPfIRE, we are committed to generating robust clinical evidence, especially for pulsed field ablation, that not only informs treatment decisions but also drives innovation in AF care. Our goal is to empower clinicians with the insights needed to enhance workflows and optimise patient outcomes.”