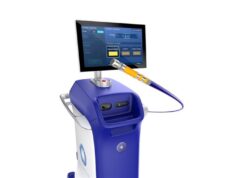
Acutus Medical has announced results from the AcQForce Flutter study, evaluating use of the AcQBlate Force sensing ablation catheter and system in the treatment of right atrial typical flutter. Results were presented during a late-breaking clinical trials and first report investigations session at the 2023 AF Symposium (2–4 February, Boston, USA).
Acutus Medical has announced results from the AcQForce Flutter study, evaluating use of the AcQBlate Force sensing ablation catheter and system in the treatment of right atrial typical flutter. Results were presented during a late-breaking clinical trials and first report investigations session at the 2023 AF Symposium (2–4 February, Boston, USA).
AcQForce Flutter Study enrolled 110 patients at 21 sites globally and was designed to evaluate the safety and efficacy of the AcQBlate FORCE sensing ablation catheter and system in the treatment of right atrial typical flutter.
Acute success was achieved in 94% of patients and there were no serious adverse events. Ablation time was 13.5±10 minutes, 33% less than comparative systems (20±14 min in BLOCK-CTI). AcQBlate required only 250.4±172.2ml of irrigation during the procedure, significantly less than comparative systems (699±386 min in BLOCK-CTI). Overall, the study demonstrated comparable outcomes to historic controls with significantly shorter ablation time and lower fluid volumes.
“AcQBlate’s novel gold tip ablation catheter performed extremely well during the clinical IDE atrial flutter cases. Short procedural times, stable contact force, and less irrigation volume contributed to efficient and safe ablation procedures. The delivery of low irrigation volumes during ablation should result in better fluid management and post-procedure patient recovery,” said Gery Tomassoni (Baptist Health Lexington, Lexington, USA), the principal investigator of the study.
The AcQBlate FORCE sensing ablation catheter and system, which received CE mark in 2020, is commercially available in Europe. Acutus submitted its premarket approval application to the US Food and Drug Administration (FDA) for the AcQBlate FORCE sensing ablation catheter and system in October 2022 and is currently under review.