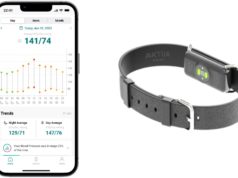
Biobeat, a global leader in wearable remote patient monitoring solutions for the healthcare continuum, announced today that its wearable remote patient monitoring devices have received 510(k) clearance from the US Food and Drug Administration (FDA) to monitor stroke volume and cardiac output in addition to cuffless blood pressure, blood oxygen saturation, pulse rate, respiratory rate and body temperature.
Biobeat’s remote patient monitoring devices are the first-ever to receive FDA clearance for cuffless blood pressure monitoring from photoplethysmography (PPG) only. The company’s cloud-based platform is designed to support health teams by transmitting real-time patient data and facilitating patient-physician interactions.
“The additional FDA clearances to monitor stroke volume and cardiac output represent our ongoing efforts to provide innovative and reliable clinical solutions to clinicians and patients in both at-home and in-hospital settings,” said Arik Ben Ishay, founder and CEO of Biobeat. “This milestone is a further validation of our proprietary non-invasive reflective PPG monitoring platform, which is already in use across a number of clinical networks in the USA.”
“Our devices are now FDA-cleared to monitor a total of seven health metrics including the five vital signs, which all medical practitioners are required to assess regardless of healthcare settings,” said Arik Eisenkraft, VP of Clinical and Regulation at Biobeat. “With these additional clearances for stroke volume and cardiac output, we expect to further equip clinicians with the necessary tools to provide fast and accurate detection that can lead to timely actionable results for patients with cardiac disease, whose needs remain severely overlooked.”