Cardiac Insight has announced the launch of the Heart@Home electrocardiogram (ECG) test kit to address new telehealth demands placed on cardiology practices by the COVID-19 response.
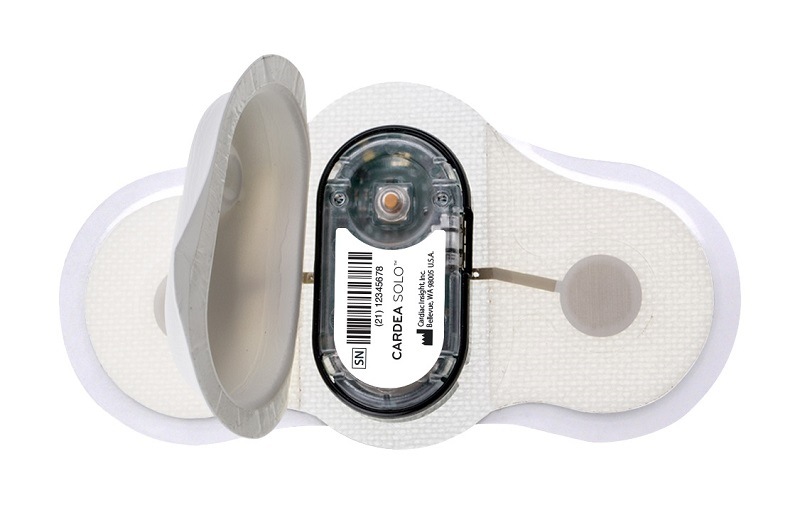
The kit enables physicians to prescribe the arrhythmia diagnosis technology of the Cardea SOLO Sensor which is express-shipped directly from Cardiac Insight to the patient’s home, along with instructions. The patient then mails the single-use sensor to the cardiologist’s office in the included shipping envelope after wearing the sensor for the prescribed time period.
Clinicians can retrieve data from the device to inform diagnoses using Cardea SOLO’s smart cable and proprietary algorithm-based ECG software, provided with the purchase of the Heart@Home ECG test kit. The process in the office takes less than five minutes to create a complete ECG report.
Robert Odell, president and chief operating officer of Cardiac Insight said, “We believe the new Heart@Home programme further strengthens the physician and patient relationship in a time when trust and privacy is more critical than ever, by fostering direct interactions and removing third-party data analysis vendors from the equation.”
The Cardea SOLO ECG System allows physicians to reduce the diagnostic timeline of patients potentially at risk of cardiac arrhythmias from weeks to just days. These arrhythmias include atrial fibrillation (AF). If left undiagnosed, AF can lead to debilitating stroke, heart failure, and sudden cardiac death.
The Cardea SOLO Heart@Home ECG Test Kit is currently available for office-based practices and hospital clinics in the USA and is designed to address the needs of electrophysiologists, cardiologists, internal medicine and family medicine physicians.








