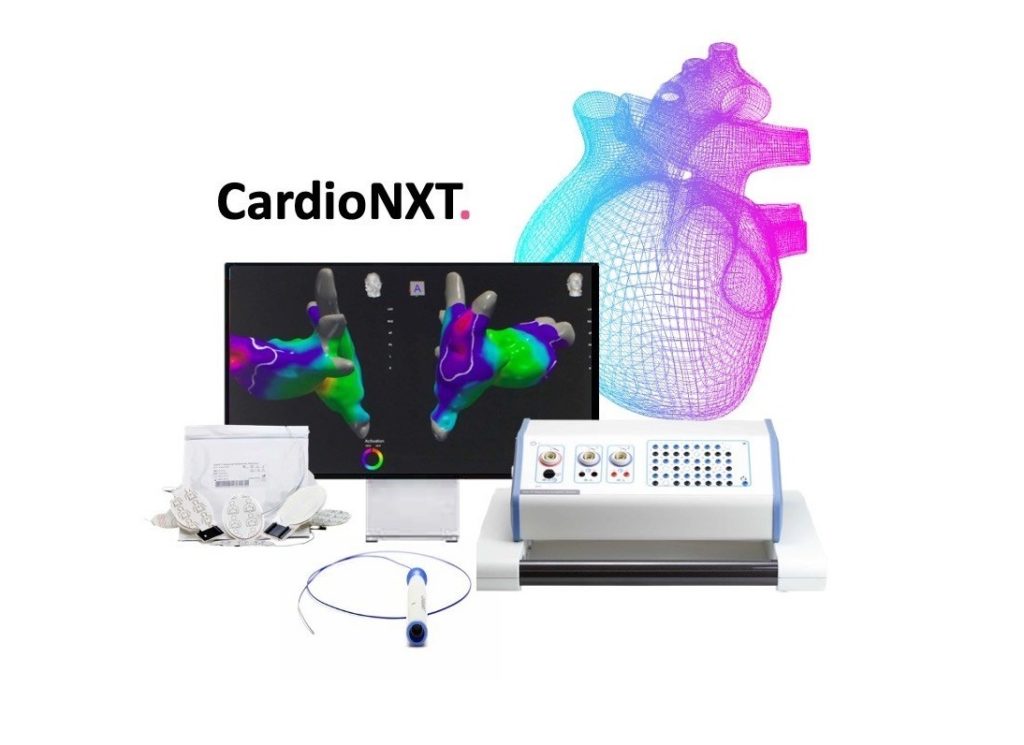
CardioNXT has received marketing clearance from the US Food and Drug Administration (FDA) for the platform technology comprised of the iMap 3D Navigation & Mapping system, Activate software, Sensor Enabled Axis Patient Patches, and MultiLink sensor enabled catheter.
The CardioNXT technology platform enables the examination of problematic cardiac tissue that is causing arrhythmias and directs the delivery of instruments to target tissue with precision.
The iMap system is the first of its kind to incorporate “dynamic referencing” based 3D navigation in cardiac procedures, CardioNXT said in a press release. While dynamic referencing is employed in various procedures for brain, spine, ear, nose and throat, and lung surgeries, it is currently not widely used in cardiac procedures.
The iMap System achieves cardiac dynamic referencing via a patented combination of dual tracking modalities, electromagnetic and impedance, which utilises for the first time a dual sensor enabled internal reference catheter, CardioNXT’s MultiLink Catheter.
“I have used products from and advised many mapping system companies. My advice to all is that there must be a tight integration between complex mapping and accurate navigation in order for any map of the heart to be useful in the treatment of patients, especially complex cases such as persistent atrial fibrillation (AF),” says Vivek Reddy, director of Cardiac Arrhythmia Service at Mount Sinai Hospital, New York, USA. “CardioNXT has taken my advice to heart. I’m excited to see the improvements that can be made in the treatment of AF patients with this technology. Treatment paradigms beyond pulmonary vein isolation (PVI) are still to be determined and we need systems like this one to help us evolve treatment strategies.”
“We were the first centre in the world to use the full combination of system, software, and catheter in human patients at Homolka Hospital,” adds Petr Neužil, chairman of Cardiology at Homolka Hospital, Prague, Czech Republic. “We are impressed with the system’s stability and accuracy and look forward to this technology being more widely available to help us treat patients with cardiac arrhythmias.”









