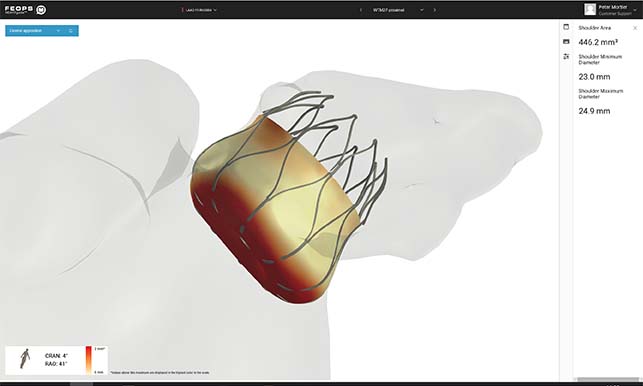
FEops has announced that it received authorisation from the US Food and Drug Administration (FDA) for FEops Heartguide pre-operative planning of left atrial appendage occlusion (LAAO) with Abbott’s Amplatzer Amulet and Boston Scientific’s Watchman FLX device.
FEops Heartguide is a cloud-based procedure planning solution in the structural heart space, based on digital twin technology. With the US introduction for the LAAO workflow, FEops Heartguide will enable US physicians to virtually model clinical scenarios with different implant positions and sizes of these FDA cleared LAA devices, helping the physician to select the optimal size and position for every individual patient.
“FEops Heartguide is the new generation preoperative planning allowing operators to enter the procedure room with increased confidence for sizing and position for LAAO interventions,” said Devi Nair (St Bernard’s Heart & Vascular Center, Jonesboro, USA). “The platform is easy to use, very intuitive and provides critical data on how the device will interact with the LAA anatomy. The software provides implanting physicians high-quality images that enhance pre-procedure planning for proper sizing of the LAAO device and implant depth. It gives the operator the best chance to choose and implant the correct device with confidence and get it right the first time.”
“FEops enabled me to select the appropriate device size and optimal device position upfront, resulting in an efficient, trouble-free, and successful ICE-guided LAAO procedure,” said Matthew Price (Scripps Clinic, La Jolla, USA).
“This FDA 510(k) clearance, following shortly after the FDA de novo clearance for FEops Heartguide LAAO workflow, is an additional milestone for FEops, allowing us to bring this innovative planning solution to the US physcians and patients for all commercially available LAAO devices,” said Matthieu De Beule, co-founder and CEO of FEops. “FEops Heartguide was already cleared for use in the European Union, UK, Canada and Australia for the LAAO and TAVI [transcatheter aortic valve implantation] workflows with more than 2,000 cases processed last year. We look forward to expand our innovative solution to the US market and streamline the way structural heart interventionions are planned.”








