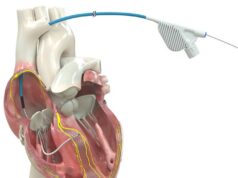
Medtronic has announced US Food and Drug Administration (FDA) clearance and commercial launch for the SelectSite C304-HIS deflectable catheter system for use in procedures involving His-bundle pacing (HBP).
According to a press release, the SelectSite C304-HIS deflectable catheter system features a deflectable, out-of-plane curve to reach the bundle of His and is designed to enable enhanced range maneuverability, fixation and implant success for a wider range of patient anatomies. Its adjustable form helps accommodate and secure the pacing lead in more challenging anatomies, including patients with a large right atrium.
“Pacing at the bundle of His is a promising procedure with a growing body of clinical evidence,” said Pugazhendhi Vijayaraman, director of electrophysiology at Geisinger Heart Institute in Wilkes-Barre, USA. “The C304-HIS catheter’s adjustable curve design should help implanters reach the His bundle fibers more easily and accommodate a variety of patient anatomies.”
The new catheter system facilitates placement of the Medtronic SelectSecure MRI SureScan Model 3830 cardiac pacing lead, the only pacing lead on the US market approved for HBP. His-bundle pacing is a physiologic alternative to right ventricular pacing, in which a pacing lead is placed at or near the bundle of His, using the heart’s native His-Purkinje conduction system to restore the heart’s normal rhythm.
“There is a growing trend of physicians interested in His-bundle pacing for certain patients as it leverages the native conduction system,” said Rob Kowal, chief medical officer, vice president of medical affairs in the Cardiac Rhythm and Heart Failure division, which is part of the Cardiac and Vascular Group at Medtronic. “Medtronic is pleased to continue to lead the industry with product innovations and training to support this emerging therapy technique.”