Patient enrolments to the BIOSync CLS randomised, controlled clinical trial have begun, according to Biotronik.
BIOSync CLS will investigate the efficacy of the uniquely physiologic rate response sensor CLS in preventing syncope.
International coordinator for the study Michele Brignole, Tigullio Hospital, Italy, comments, “Previous studies have provided contradictory results regarding the benefit of cardiac pacing in syncope, and the current European Society of Cardiology (ESC) guidelines for this indication suggest that further study is necessary. The BIOSync CLS trial has the potential to clarify whether a dual-chamber pacemaker with the uniquely physiologic CLS pacing algorithm is able to detect and quickly neutralise syncope. I am very excited to begin this highly significant trial.”
The international, double-blinded multicentre trial will include 30 study centres in Italy, Spain, France, the Netherlands, Portugal, and Canada.
Investigators will randomise 128 patients with a pacemaker indication for recurrent syncope into two groups. Patients in each study arm will receive a Biotronik dual-chamber pacemaker with CLS turned either on or off. The trial’s primary endpoint is to compare the time to first recurrence of syncope between the active group (detection mode and CLS turned on) and the control group (placebo, detection mode only). There will be a follow-up period of two years; information regarding primary endpoint events will be evaluated via anonymous patient questionnaires gathered by independent personnel blinded to the randomisation.
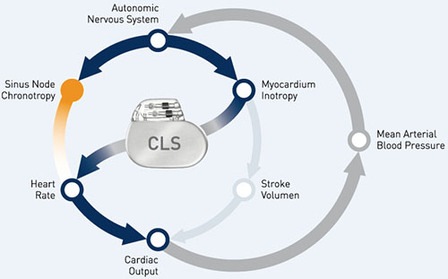
“A number of previous trials have shown that CLS is sensitive to contractility. We hypothesise that pacemakers with CLS are uniquely capable of detecting the increase in contractility which can occur in the early stage of a syncopal event. They should therefore be able to initiate cardiac pacing at an earlier stage to prevent the sudden drop in heart rate which often causes syncope,” states first implanter Marco Tomaino, Bolzano Hospital, Italy. “If our hypothesis is proven correct, implications for treatment indications and guidelines would be widespread.”









