Researchers have used an optical mapping system to observe how the malaria drug hydroxychloroquine, which has been touted as a possible treatment for COVID-19, creates disturbances in heart rhythm in an animal model. The research, published in the journal Heart Rhythm, found that the drug made it “surprisingly easy” to trigger arrhythmias in two types of animal hearts by altering the timing of electrical waves that control heartbeat.
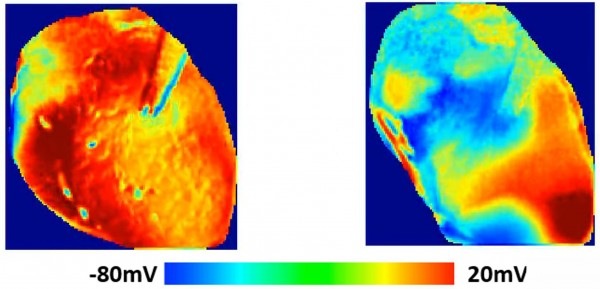
“We have illustrated experimentally how the drug actually changes the waves in the heart, and how that can initiate an arrhythmia,” said Flavio Fenton, a professor in the School of Physics at the Georgia Institute of Technology (Atlanta, USA) and the paper’s corresponding author. “We have demonstrated that with optical mapping, which allows us to see exactly how the waveform is changing. This gives us a visual demonstration of how the drug can alter the wave propagation in the heart.”
The team observed an elongation of the T wave, noting that by extending the QT portion of one wave cycle, the drug sets the stage for disturbances in the next wave, potentially creating an arrhythmia. Such disturbances can transition to fibrillation.
The ability to easily trigger disturbances known as “long QT” reinforces cautions about using hydroxychloroquine in humans – particularly in those who may have heart damage from Covid-19, cautioned Shahriar Iravanian, a co-author of the paper and a cardiologist in the Division of Cardiology, Section of Electrophysiology, at Emory University Hospital (Atlanta, USA).
“The hearts used in the study are small and very resistant to this form of arrhythmia,” Iravanian said. “If we had not seen any HCQ-induced arrhythmias in this model, the results would not have been reassuring. However, in reality, we observed that hydroxychloroquine readily induced arrhythmia in those hearts. This finding is very concerning and, in combination with the clinical reports of sudden death and arrhythmia in COVID-19 patients taking hydroxychloroquine, suggests that the drug should be considered a potentially harmful medication and its use in COVID-19 patients be restricted to clinical trial settings.”
Georgia Tech postdoctoral fellow Ilija Uzelac administered hydroxychloroquine to the animal hearts—one from a guinea pig and one from a rabbit—while quantifying wave patterns changing across the hearts using a high-powered, LED-based optical mapping system. Voltage-sensitive fluorescent dyes made the electrical waves visible as they moved across the surface of the hearts.
“The effect of the arrhythmia and the long QT was quite obvious,” said Uzelac. “hydroxychloroquine shifts the wavelengths to larger values, and when we quantified the dispersion of the electrical current in portions of the heart, we saw the extension of the voltage across the tissue. The change was very dramatic comparing the waveforms in the heart with and without the hydroxychloroquine.”
The drug concentration used in the study was at the high end of what is being recommended for humans. hydroxychloroquine normally takes a few days to accumulate in the body, so the researchers used a higher initial dose to simulate the drug’s effect over time.
Patients taking hydroxychloroquine for diseases such as lupus and rheumatoid arthritis rarely suffer from arrythmia because the doses they take are smaller than those being recommended for COVID-19 patients, Iravanian said.
“COVID-19 patients are different and are at a much higher risk of hydroxychloroquine-induced arrhythmia,” he said. “Not only is the proposed dose of hydroxychloroquine for COVID-19 patients two to three times the usual dose, but COVID-19 has effects on the heart and lowers potassium levels, further increasing the risk of arrythmias.”
Fenton and colleagues have begun a new study to evaluate the effects of hydroxychloroquine with the antibiotic azithromycin, which has been suggested as a companion treatment. Azithromycin can also cause the long QT effect, potentially increasing the impact on COVID-19 patients.
The study, which was supported by grants from the National Institutes of Health and National Science Foundation, was also coauthored by Hiroshi Ashikaga from Johns Hopkins University School of Medicine (Baltimore, USA); Neal Bathia from the Division of Cardiology, Section of Electrophysiology at Emory University Hospital (Atlanta, USA); Conner Herndon, Abouzar Kaboudian, and James C Gumbart from the Georgia Tech School of Physics (Atlanta, USA), and Elizabeth Cherry from the Georgia Tech School of Computational Science and Engineering (Atlanta, USA).