Tag: Protaryx
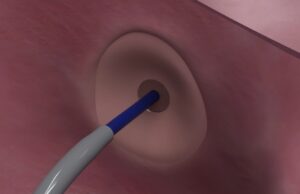
Protaryx Medical receives US FDA clearance for proprietary transseptal puncture device
Protaryx Medical has received 510(k) clearance from the US Food and Drug Administration (FDA) to market its proprietary transseptal puncture device.
According to the company,...
First-in-human study of Protaryx transseptal puncture technology completed
Protaryx Medical has announced the completion of its first-in-human (FIH) study, assessing its advanced transseptal puncture technology.
Conducted on 3 December 2024 at Sanatorio Italiano...