Malcom Finlay (Bart’s Health Centre, London, UK) reviews the growing role of artificial intelligence (AI) in diagnosing arrhythmias.
Johnny, a 72-year-old film producer, is meeting clients when his watch gently vibrates notifying him of his irregular pulse. He pressed his finger to his strap, and with a swipe sends the single lead ECG to his cardiologist without pausing the meeting. The AI delivered backbone powering self-diagnostics and wearables is invisible but always present: AI is taking over the medical world. Or so it would seem.
What real benefits and implications are there of AI for the hospital treatment of arrhythmia patients? It is one thing to triumph self-screening with an efficient Silicon Valley powered gizmo for the “worried well”, but quite another for AI to leverage the highly-toned skills of the cardiologist—let alone the electrophysiologist.
The “AI” buzz has affected several technologies and disciplines during past decade, the current darling of this buzz is “deep learning” (itself a subset of machine learning). Data are passed across multiple layers (hence “deep”) of computational nodes, with processing at each level. Patterns within the data are discovered, reinforced, and algorithms are adjusted dependent on the output—i.e. data is passed back as well as forward and algorithms modified to improve performance. This can be thought of analogous to neurological learning (aka neural networks); positive and negative reinforcement drives recognition and behaviour changes within the model.
AI processes are extremely computationally intensive and although the underlying theories are several decades old, the exponential acceleration of the field has been thanks to the ubiquitous and widespread availability of super-computing (think Amazon Web Services) and data availability (think social media). Serious excitement comes from the effectiveness at learning from unsorted bulk data, and enabling insights without prespecifying output groupings. Hence these methods can become extremely good at, for instance, labelling pictures of cats without actually knowing what a “cat” is.
In fact, AI encompasses far more than just deep learning.
State-of-the-art techniques combine both machine learning and statistics, such as multi-level regression or “k-means classification”. Classical AI, oft forgotten but extremely powerful, chiefly refers to algorithms performing rule-based analytics from known data structures. The forefront of AI implementations—i.e. what gets investors really sitting up—is the combination these techniques together. Ultimately, this leverages distinct advantages of each method to produce far more useful and robust analytical results.
The AI hype in arrhythmia has been “you could take a bunch of ECGs, put it through AI and get the answer”, be it in diagnosis, prognosis, or treatment targeting. Unfortunately, the Achilles heel here is the frustratingly poor structures and classification of our data. Diagnostic classifications are loose (my atrial fibrillation is not the same as your atrial fibrillation), data structures poor, follow-up inconsistent and, critically, outcomes classifications are weak. In healthcare, we are (mostly) trying to accomplish more than “label pictures of cats” and arrhythmia is a difficult problem indeed.
 The number one issue with training AI systems comes from the data; the old line “rubbish in, rubbish out” is as true with machine learning as with any computer science process. And we do not need much “rubbish”—misclassified ECGs, incorrect patient diagnoses or serendipitous improvements unrelated to recorded treatments—to significantly reduce machine learning performance. An AI system may be near perfect on a training dataset, yet hopeless in the real world. Compounding this are (rightly) stringent data handling regulations, and unlike consumer-grade devices, harvested patient data cannot be used to continually improve performance of our systems. Even if this is circumvented, current interpretations of strict medical device regulations stall dissemination of diagnostic or guiding treatment.
The number one issue with training AI systems comes from the data; the old line “rubbish in, rubbish out” is as true with machine learning as with any computer science process. And we do not need much “rubbish”—misclassified ECGs, incorrect patient diagnoses or serendipitous improvements unrelated to recorded treatments—to significantly reduce machine learning performance. An AI system may be near perfect on a training dataset, yet hopeless in the real world. Compounding this are (rightly) stringent data handling regulations, and unlike consumer-grade devices, harvested patient data cannot be used to continually improve performance of our systems. Even if this is circumvented, current interpretations of strict medical device regulations stall dissemination of diagnostic or guiding treatment.
So how will medical-grade AI systems change the lives of the humble electrophysiologist? The mass self-screening for atrial fibrillation by wearables powered by AI will sweep through our clinics. If your waiting list is short—it soon will not be. AI will have its presence felt across the hospital, increasing the “efficiencies” of purchasing, lab list booking and so on. Much of the cath lab hardware will become “intelligent” (think automatic adjustment of fluoro settings and table positions during cases), though it will be a while before we can have a chat about politics with the image intensifier.
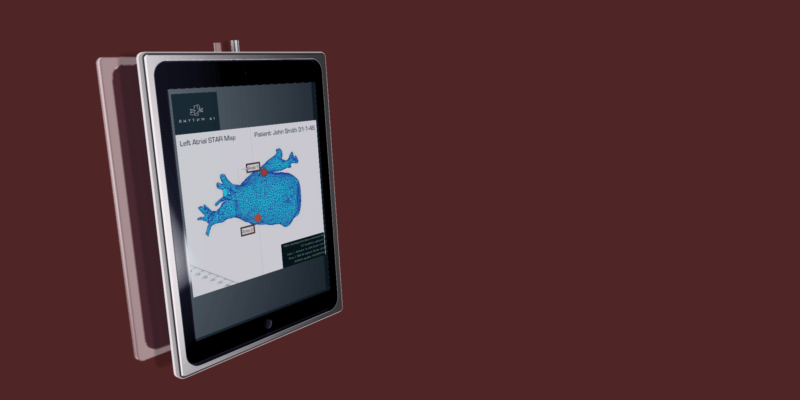
The “hard” stuff we do will be made significantly easier, perhaps most obviously interpretation of complex mapping data. This is the underlying aim of Rhythm AI’s STAR mapping system. Here, we leveraged a combination of painstakingly curated patient data with algorithmic and machine learning AI to identify sites driving persistence of atrial fibrillation on intra-cardiac mapping. Preliminary clinical data are very promising, and a multicentre clinical trial will assess formal outcomes against state-of-the-art ablation techniques.
Despite Johnny’s fancy watch, AI is far, far away from replacing the electrophysiologist. Some low level advice could seem plausible for such a system to give (“Lose weight! Exercise more! Take an anticoagulant!”), yet this is the fringe of the electrophysiologist’s practice. Our role is as a trusted advocate, providing balanced and individualised personal recommendations. The (thus far) failure of robotics to make serious inroads into the electrophysiologist lab also points to the complexities in offloading treatment delivery to intelligent systems. For the near future such systems will still require direct physician supervision. Amongst the earliest inroads, we will notice will be a step change in the interpretation of the complex electroanatomical maps, exactly the niche STAR mapping seeks to fill.
Our world will be changed because of AI, with better patient screening, easier to interpret Electroanatomic maps and lower X-ray doses. But we should not hang up our leads just yet, hard-earned electrogram interpretation and ablation skills will not go to waste. The gulf between AI helping professional efficiency and doing a large majority of our jobs for us is a large one. As Johnny will testify, there is a long way between labelling cats on “Insta” and performing a successful ventricular arrhythmia ablation.
Malcolm Finlay is the founder and non-executive director of Rhythm AI. He is also at William Harvey Research Institute, Barts and The London School of Medicine and Dentistry, Queen Mary University of London, London, UK. STAR Mapping is an investigational device and is not currently approved for commercial sale.