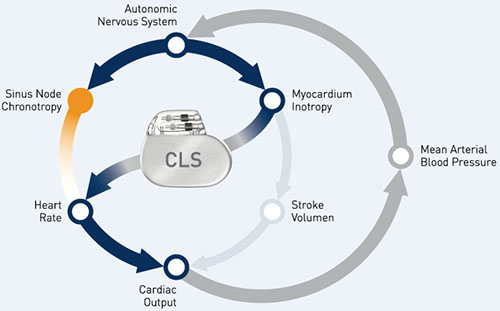
The first patient enrolments to the randomised, controlled B3 clinical trial have begun, according to Biotronik. The B3 trial will investigate the unique physiologic rate response sensor Closed Loop Stimulation (CLS). It will evaluate the potential benefit of this device for patients with atrial fibrillation (AF). The study will address the question of whether pacemakers and implantable cardioverter-defibrillators (ICDs) with CLS can reduce the rate of stroke.
Valeria Calvi, first implanter and international coordinator for the study, University of Catania, Ferrarotto Hospital, Italy, comments, “I am very excited to begin this trial. We hope to demonstrate that the physiological heart rate modulation induced by CLS in sick sinus disease patients can control clinically relevant AF episodes. CLS may well improve clinical outcomes and lower the incidence of stroke in this particularly challenging patient population.”
The large multicentre trial will include 52 study centres in Italy, Spain, China, Taiwan, Korea, Singapore, Malaysia and India. Investigators will randomise 1,308 patients with a pacemaker or ICD indication who require dual-chamber pacing due to sinus node disorder (SND) into two groups. Patients in each study arm are to receive a Biotronik pacemaker or ICD with CLS turned either on or off. The trial’s primary endpoint is the time to first onset of new clinically relevant AF episodes or thromboembolic events, and there will be a follow-up period of three years. As a secondary endpoint, the study will investigate whether a subgroup of patients remotely monitored using Biotronik Home Monitoring is associated with an even further reduced risk of stroke and persistent AF.
“Two previous studies have already demonstrated a significantly lower atrial burden for patients with CLS pacemakers. We are eager to learn how CLS might reduce the risk of clinically relevant AF and stroke, and improve patients´ quality of life,” says Klaus Contzen, director of the center for clinical research at Biotronik.









