
Jonathan Behar (London, UK) co-inventor for the Guide CRT platform—shortlisted as a 2017 finalist for the EHRA Inventors Award—details the benefits of this novel technology, which enables the real-time analysis and fusion of cardiac magnetic resonance imaging (MRI)-derived scar and dyssynchrony data to guide LV lead implantation in cardiac resynchronisation therapy.
Cardiomyopathies can cause electrical disturbance and slow myocardial conduction, resulting in a delay between left and right ventricular activation, most often seen as LBBB.1 A subsequent disco-ordinated, inefficient biventricular contraction results in wasted energy, impaired cardiac output2 and progressive left ventricular dilatation3 leading to significant morbidity and mortality. Biventricular pacing or cardiac resynchronisation therapy (CRT) is a highly effective electrical therapy for selected heart failure patients with persistent symptoms despite optimal medical therapy, prolonged QRS duration and a severely depressed left ventricular (LV) systolic function. It is thought to act by restoring electro-mechanical synchrony.4
Suboptimal response to CRT
Despite delivering CRT for over two decades, a significant proportion (30‒50%) fail to improve as demonstrated in the 2012 EHRA survey of CRT.5 Disappointingly, this proportion has remained static over a period of extensive technology and technique development. Causes of suboptimal CRT response are multi-factorial and include suboptimal LV lead placement, ineffective electro-mechanical resynchronisation, untreated anaemia and ineffectual atrioventricular (AV) and ventriculoventricular (VV) timings with respect to device optimisation.6
The evidence-base for image guidance
An increasing number of clinical studies have demonstrated the importance of avoiding areas of myocardial fibrosis and targeting regions of latest mechanical activation or greatest dyssynchrony, as an approach to identifying a target for LV lead placement. TARGET and STARTER were both single-centre randomised controlled studies evaluating the use of speckle tracking echocardiography for pre procedural targeting of myocardial segments for LV lead delivery, as compared with conventional non-image guided implantation.7,8 Patients in whom an LV lead was placed in a region of latest mechanical activation had significantly reduced rates of heart failure hospitalisation and death compared with those where the lead was distant. A large retrospective registry of 559 patients in whom pre procedural cardiac MRI was performed, demonstrated that patients with the LV lead implanted in areas of myocardial fibrosis had a greater hazard ratio of cardiovascular death or heart failure hospitalisation compared with those where the lead was out of scar (HR 5.57, p<0.0001). More recently, a selection of multi-modality imaging studies have shown the additional benefit for avoiding scar and targeting dyssynchrony amongst patients being implanted with CRT.9‒11 A common theme amongst these studies is the observed improvement in CRT response rate (approximately 15‒20%) in those implanted using an image-guided approach.
Real-time image fusion to guide CRT
Whilst the results from the growing number of image guidance studies is encouraging, one of the major limitations is that the data output from the imaging modality in each study is reviewed separate from, and alongside, X-ray rather than integrated together. Given the radiolucency of the cardiac silhouette and high variability in the rotation of the left and right-sided chambers relative to one another, it is not altogether surprising that using fluoroscopy to determine regional anatomy can be highly inaccurate, particularly with regard to CRT and lead position.12
Earlier this year, our group published the first clinical study to evaluate a platform (Guide CRT) which enables the real-time analysis and fusion of cardiac MRI-derived scar and dyssynchrony data to guide LV lead implantation.13 Upon completion of a routine cardiac MRI scar, the patient is transferred to the adjacent cardiac catheter lab (X-MRI suite) whilst simultaneously, the imaging dataset is processed within 25 minutes. Segmentation of long and short axis MRI sequences generates a 3-dimensional (3D) mesh and the Guide CRT display is shown detailing the location, burden and transmurality of myocardial fibrosis, in addition to volume vs. time curves for the 16-American Heart Association (AHA) segments, in order to identify regions of greatest dyssynchrony (Figure 1).

Following a short registration step, the 3D model is registered to the X-ray co-ordinate system and crucially, any subsequent X-ray acquisitions during the case are displayed with instantaneous overlay of the correctly oriented 3D model. Upon coronary venography, the 3D derived model of the patient’s left ventricle is instantaneously fused enabling the implanting physician to identify the patient specific target locations for LV lead placement and how these are subtended by the coronary venous tree (Figure 2).
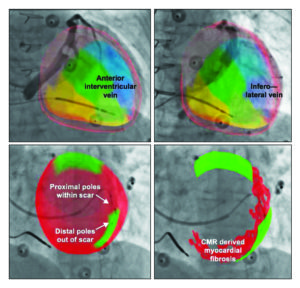
This platform was safely tested in 14 patients with conventional indications for CRT. It demonstrated that pacing with cardiac MRI-derived segments out of scar had more favourable electrical properties (lower capture thresholds – mean difference 2.4 Volts (1.5-3.2), p<0.001 and lower paced QRS duration – mean difference 25ms (15‒34), p<0.001) as compared with pacing within cardiac MRI-derived regions of scar). The mixed effect model analysis validated the accuracy of the cardiac MRI-derived, fused 3D model.
Personalised therapy for the 21st century
Empirical placement of the LV lead in the inferolateral wall has been the standard of care for the past two decades of CRT implantation and yet there are clear data to show the optimal LV site is highly patient specific.14 Utilising the individual’s coronary venous anatomy, scar distribution, electrophysiology and mechanical contraction patterns to identify and target the optimal site for the LV lead is one novel approach toward improving the effectiveness of CRT. Larger scale, multicentre, randomised clinical trials will be required to demonstrate whether this approach yields greater CRT response compared with the conventional implantation method.
The recent emergence of LV endocardial stimulation as an alternative route to delivering biventricular pacing for CRT has a growing evidence base15 with some data showing superior haemodynamic and electrophysiological indicies16‒19. The use of the image guidance strategies described may be particularly suitable for this approach with the ability to truly target any region on the LV endocardial wall without the constraint of the coronary venous anatomy. Recent development of a wireless intracardiac LV endocardial electrode for CRT delivery (WiSE-CRT, EBR systems) represents a unique opportunity to use integrated multi modality image guidance for optimal LV site selection (Figure 3).20

In the future, refinements in hybrid catheter lab technology will enable the seamless integration of cardiac imaging data with X-ray, providing a visualisation scaffold to improve physician confidence in accurate pacemaker lead deployment. Real-time image fusion is rapidly becoming a reality for an increasing number of cardiac centres; a more streamlined workflow and improvement in accuracy and registration will enable this technology to be used in more translational clinical work in the future.
References:
- Grines CL, Bashore TM, Boudoulas H, Olson S, Shafer P, Wooley CF. Functional abnormalities in isolated left bundle branch block. The effect of interventricular asynchrony. Circulation [Internet]. American Heart Association Journals; 1989 Apr [cited 2016 Jul 11];79(4):845–53. Available from: http://www.ncbi.nlm.nih.gov/pubmed/2924415
- Prinzen FW, Augustijn CH, Arts T, Allessie MA, Reneman RS. Redistribution of myocardial fiber strain and blood flow by asynchronous activation. Am J Physiol [Internet]. 1990 Aug [cited 2016 Jul 10];259(2 Pt 2):H300-8. Available from: http://www.ncbi.nlm.nih.gov/pubmed/2386214
- Abraham WT, Hayes DL. Cardiac resynchronization therapy for heart failure. Circulation [Internet]. American Heart Association Journals; 2003 Nov 25 [cited 2016 Jul 10];108(21):2596–603. Available from: http://www.ncbi.nlm.nih.gov/pubmed/14638522
- Cazeau S, Ritter P, Bakdach S, Lazarus A, Limousin M, Henao L, et al. Four chamber pacing in dilated cardiomyopathy. Pacing Clin Electrophysiol [Internet]. Blackwell Publishing Ltd; 1994 Nov [cited 2016 Jul 10];17(11):1974–9. Available from: http://doi.wiley.com/10.1111/j.1540-8159.1994.tb03783.x
- Daubert J-C, Saxon L, Adamson PB, Auricchio A, Berger RD, Beshai JF, et al. 2012 EHRA/HRS expert consensus statement on cardiac resynchronization therapy in heart failure: implant and follow-up recommendations and management. Europace [Internet]. 2012 Sep [cited 2015 Jan 7];14(9):1236–86. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22930717
- Mullens W, Grimm R a, Verga T, Dresing T, Starling RC, Wilkoff BL, et al. Insights from a cardiac resynchronization optimization clinic as part of a heart failure disease management program. J Am Coll Cardiol [Internet]. American College of Cardiology Foundation; 2009 Mar 3 [cited 2014 Nov 16];53(9):765–73. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19245967
- Khan FZ, Virdee MS, Palmer CR, Pugh PJ, O’Halloran D, Elsik M, et al. Targeted left ventricular lead placement to guide cardiac resynchronization therapy: the TARGET study: a randomized, controlled trial. J Am Coll Cardiol [Internet]. Elsevier Inc.; 2012 Apr 24 [cited 2014 Nov 12];59(17):1509–18. Available from: http://www.ncbi.nlm.nih.gov/pubmed/22405632
- Saba S, Marek J, Schwartzman D, Jain S, Adelstein E, White P, et al. Echocardiography-guided left ventricular lead placement for cardiac resynchronization therapy: results of the Speckle tracking assisted resynchronization therapy for electrode region trial. Circ Heart Fail [Internet]. 2013 May [cited 2014 Nov 15];6(3):427–34. Available from: http://www.ncbi.nlm.nih.gov/pubmed/23476053
- Bertini M, Mele D, Malagù M, Fiorencis A, Toselli T, Casadei F, et al. Cardiac resynchronization therapy guided by multimodality cardiac imaging. Eur J Heart Fail [Internet]. John Wiley & Sons, Ltd; 2016 Nov [cited 2016 Nov 6];18(11):1375–82. Available from: http://doi.wiley.com/10.1002/ejhf.605
- Sommer A, Kronborg MB, Nørgaard BL, Poulsen SH, Bouchelouche K, Böttcher M, et al. Multimodality imaging-guided left ventricular lead placement in cardiac resynchronization therapy: A randomized controlled trial. European Journal of Heart Failure. 2016; 18(11):1365‒1374
- Nguyên UC, Mafi-Rad M, Aben J-P, Smulders MW, Engels EB, van Stipdonk AMW, et al. A novel approach for left ventricular lead placement in cardiac resynchronization therapy: Intra-procedural integration of coronary venous electro-anatomic mapping with delayed enhancement cardiac magnetic resonance imaging. Heart Rhythm [Internet]. 2016 Sep 20 [cited 2016 Oct 5]; Available from: http://www.ncbi.nlm.nih.gov/pubmed/27663606
- Sommer A, Kronborg MB, Nørgaard BL, Gerdes C, Mortensen PT, Nielsen JC. Left and right ventricular lead positions are imprecisely determined by fluoroscopy in cardiac resynchronization therapy: a comparison with cardiac computed tomography. Europace [Internet]. 2014 Sep [cited 2014 Nov 16];16(9):1334–41. Available from: http://www.ncbi.nlm.nih.gov/pubmed/24687965
- Behar JM, Mountney P, Toth D, Reiml S, Panayiotou M, Brost A, et al. Real-Time X-MRI-guided left ventricular lead implantation for targeted delivery of cardiac resynchronization therapy. JACC Clin Electrophysiol [Internet]. 2017 Apr [cited 2017 Jul 9]; Available from: http://www.sciencedirect.com/science/article/pii/S2405500X17301305
- Derval N, Steendijk P, Gula LJ, Deplagne A, Laborderie J, Sacher F, et al. Optimizing hemodynamics in heart failure patients by systematic screening of left ventricular pacing sites: the lateral left ventricular wall and the coronary sinus are rarely the best sites. J Am Coll Cardiol [Internet]. Elsevier Inc.; 2010 Feb 9 [cited 2014 Nov 14];55(6):566–75. Available from: http://www.ncbi.nlm.nih.gov/pubmed/19931364
- Morgan JM, Biffi M, Gellér L, Leclercq C, Ruffa F, Tung S, et al. Alternate site cardiac resynchronization (ALSYNC): a prospective and multicentre study of left ventricular endocardial pacing for cardiac resynchronization therapy. Eur Heart J [Internet]. 2016;ehv723. Available from: http://eurheartj.oxfordjournals.org/lookup/doi/10.1093/eurheartj/ehv723
16. Behar JM, Jackson T, Hyde E, Claridge S, Gill J, Bostock J, et al. Optimized left ventricular endocardial stimulation is superior to optimized epicardial stimulation in ischemic patients with poor response to cardiac resynchronization therapy: A combined magnetic resonance imaging, electroanatomic contact mapping, and he. JACC Clin Electrophysiol. 2016;2(7)
17. Bordachar P, Derval N, Ploux S, Garrigue S, Ritter P, Haissaguerre M, et al. Left ventricular endocardial stimulation for severe heart failure. J Am Coll Cardiol [Internet]. Elsevier Inc.; 2010;56(10):747–53. Available from: http://linkinghub.elsevier.com/retrieve/pii/S0735109710023302
18. Hyde ER, Behar JM, Claridge S, Jackson T, Lee AWC, Remme EW, et al. Beneficial effect on cardiac resynchronization from left ventricular endocardial pacing is mediated by early access to high conduction velocity tissue: Electrophysiological simulation study. Circ Arrhythmia Electrophysiol 2015;8(5)
19. Prinzen FW, Van Deursen C, Van Geldorp IE, Rademakers LM, Van Hunnik A, Kuiper M, et al. Left ventricular endocardial pacing improves resynchronization therapy in canine left bundle-branch hearts. Circ Arrhythmia Electrophysiol 2009;2(5):580–7
20. Behar JM, Sieniewicz B, Mountney P, Toth D, Panayiotou M, Claridge S, et al. Image integration to guide wireless endocardial LV electrode implantation for CRT [Internet]. JACC: Cardiovascular Imaging. 2017 [cited 2017 Jul 9]. Available from: http://www.sciencedirect.com/science/article/pii/S1936878X17301432
Jonathan Behar is a cardiology registrar at Barts Heart Centre, London, UK. He is a registered co-inventor for the Guide CRT platform described above. The Guide CRT platform has been submitted to obtain European patent









