dawnpowell
COVID-19: NCDR Chest Pain-MI, CathPCI registries to review effect of virus...
The American College of Cardiology (ACC) is to collect COVID-19 data through the NCDR Chest Pain-MI and CathPCI registries with the aim of capturing...
COVID-19: Anticoagulants may improve survival among hospitalised COVID patients
Corresponding author Valentin Fuster (Mount Sinai Heart, New York, USA) and colleagues report in the Journal of the American College of Cardiology that hospitalised...
COVID-19: ESC Congress 2020 to become a completely virtual event
The European Society of Cardiology (ESC) is for the first time in its 70-year history, to hold its annual meeting (ESC Congress) 100% online....
COVID-19: North American societies provide advice on resuming cardiovascular procedures
The American College of Cardiology (ACC) and other North American cardiology societies, including the Canadian Association of Interventional Cardiology (CAIC), have issued guidance on...
COVID-19: Calls for physicians to address domestic violence during social distancing
Prism Schneider (Cumming School of Medicine, University of Calgary, Calgary, Canada) and others write in a commentary in the Canadian Medical Association Journal—because of...
COVID-19: CMS outlines recommendations for restarting non-emergent procedures
On 18 March, the Centers for Medicare & Medicaid Services (CMS) recommended “limiting non-essential care and expanding surge capacity into ambulatory surgical centres and...
COVID-19: Testing for high-sensitivity cardiac troponin would be beneficial not a...
In response to a recent American College of Cardiology (ACC) review that advocated that troponin should only be measured when a diagnosis of acute...
COVID-19: How the Egyptian cardiology departments are managing the pandemic
The COVID-19 pandemic is hitting the world hard. Egypt, the most populous country in the Middle East and North Africa (MENA) region, is no...
COVID-19: American Heart Association issue pandemic CPR guidelines
The American Heart Association (AHA) has compiled interim CPR guidelines to help rescuers treat cardiac arrest patients with suspected or confirmed COVID-19. The guidelines...
COVID-19: Cotton and surgical masks may be ineffective at stopping spread...
In a brief report in Annals of Internal Medicine, Seongman Bae (Asan Medical Center, University of Ulsan College of Medicine, Seoul, Korea) and colleagues report that neither surgical...
COVID-19: Optimising heart failure management in the patient’s home
Physicians already recognised the importance of remote management for heart failure patients long before the COVID-19 pandemic. However, the current situation has heightened the...
COVID-19: European Commission agrees to postpone new MDR because of pandemic
The European Commission (EC) has adopted a proposal to postpone by one year the date of application of the new Medical Devices Regulation (MDR), which...
COVID-19: European Commission looking to postpone new MDR by one year
During a Q&A section of a European Commission (EC) college meeting on 25 March, EC spokesperson Stefan de Keersmaecker stated that the commission were looking to...
AF Symposium 2020: Cryoablation pulmonary vein isolation is safe and effective...
Hugh Calkins (Johns Hopkins Medical Institutions, Baltimore, USA) told delegates attending the AF Symposium 2020 (23–25 January, Washington, DC, USA) that cryoablation with the...
AF Symposium 2020: Myocardial specificity of pulsed field ablation provides safety...
At the AF Symposium 2020 (23–25 January, Washington, DC, USA), Vivek Y Reddy (Helmsley Electrophysiology Center, Icahn School of Medicine at Mount Sinai, New York, USA)...
AF Symposium 2020: Use-as-needed NOAC strategy is safe and effective for...
Speaking today at the AF Symposium (23–25 January, Washington, DC, USA), Francis Marchlinski (Hospital of the University of Pennsylvania, Philadelphia, USA) reported that selected...
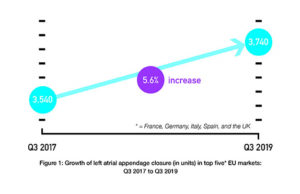
BIBA Briefings: Longest and largest follow-up data for Watchman supports its...
Data from the CAP and CAP2 registries—which contain the longest and largest follow-up of patients—add to previous evidence that left atrial appendage closure (LAAC)...
AtriCure agrees to buy LARIAT developer SentreHEART
AtriCure has entered into a definitive agreement to acquire SentreHEART, which developed the LARIAT device for left atrial appendage closure in patients with atrial...
ESC 2019: Registry study supports use of ICDs for primary prevention...
New data from the Swedish Heart Failure Registry indicate the use of an implantable cardioverter defibrillator (ICD) in patients with heart failure with reduced...
ESC 2019: Left atrial appendage closure is non-inferior to NOACS for...
At the 2019 European Society of Cardiology (ESC) Congress (31 August–4 September, Paris, France), Pavel Osmancik (Charles University and University Hospital Kralovske Vinohrady, Prague,...
Zoll Medical acquires TherOx
Zoll Medical, an Asahi Kasei Group Company that manufactures medical devices and related software solutions, has acquired TherOx—a company on improving treatment of acute...
Philips announces new advanced automation capabilities for its EPIQ CVx and...
A Philips press release reports both the EPIQ CVx and EPIQ CVxi now include automated applications for 2D assessment of the heart, as well...
First-in-human study demonstrates safety and efficacy of QDOT MICRO ablation catheter
Data presented at Heart Rhythm 2019 (8–11 May, San Francisco, USA) and published in JACC: Clinical Electrophysiology indicates that the QDOT MICRO (Biosense Webster),...
US$170m funding for Acutus Medical
A company providing what a press release calls “next-generation electrophysiology technology solutions” has announced that it now has US$170m financing. The press release reports...
EuroPCR 2019: Renal denervation linked to reduction in subclinical atrial fibrillation
Data presented at EuroPCR today indicates that renal denervation with the Symplicity system (Medtronic) is associated with a reduction in subclinical atrial fibrillation in...
Targeted blood pressure management helps to reduce strokes in patients with...
An analysis on the impact of stroke severity in patients receiving the HeartWare HVAD system (Medtronic) as destination therapy shows that targeted blood pressure...
CE mark for the FEops HEARTguide
FEops has received CE mark approval for its FEops HEARTguide. According to a press release, FEops HEARTguide is a one-in-its-kind procedure planning environment for...
We need to embrace digital education and adapt it to improve...
Recent years have seen many advances in education for cardiologists, but little attention has been paid to the impact of new technologies, particularly digital...
First trial of in-heart micro device for left atrial pressure monitoring...
Vectorious Medical Technologies has announced the initiation of the VECTOR-HF first-in-human (FIH) clinical trial, and the successful first “in-human” implantation of the V-LAPTM monitoring...
Editors-in-chief of major cardiovascular journals claim medical misinformation puts “lives at...
The editors-in-chief of major cardiovascular journals—of both US and European societies—have come together to “sound the alarm” about the dangers of medical misinformation that...
The British Heart Foundation’s “Big Beat Challenge” is now accepting proposals
The British Heart Foundation (BHF) has announced that it is now accepting proposals for its Big Beat Challenge—a single funding award of £30 million. The...
More work needed to “untangle reasons” for gender gap in cardiovascular...
Writing in Circulation: Cardiovascular Quality and Outcomes, David Ouyang (Stanford University, Falk Research Center, Palo Alto, USA) and others report that while the number...
BioCardia submits 510(k) application to US FDA for its Avance steerable...
BioCardia has announced its 510(k) submission for US FDA clearance of the Avance steerable introducer, which is designed for introducing various cardiovascular catheters into...
The first 60 years of cardiac pacing
Sixty years ago, the first pacemaker implant was made in Stockholm, Sweden (Senning & Elmqvist) on 8 October 1958. The patient had atrioventricular block...
First patient treated in STELLAR US study evaluating next-generation balloon ablation...
Biosense Webster has enrolled and treated the first patient in its STELLAR US investigational device exemption (IDE) study. The study will evaluate the safety...
CardioFocus completes enrolment in HeartLight X3 trial
CardioFocus has completed enrolment in a trial to evaluate its next-generation HeartLight X3 endoscopic ablation system. According to a press release, a total of...
“No serious adverse outcomes” with ICD shocks in young athletes playing...
A post-hoc analysis of the ICD Sports Registry—which has already indicated that many patients with implantable cardioverter defibrillators (ICD) can safely participate in vigorous...
AHA 2018: Computerised-decision support has the potential to be a “powerful...
Data presented at the 2018 American Heart Association (AHA) scientific session scientific sessions (10–12 November, Chicago, USA) indicate that the use of alert-based computerised-decision...
Presence of left bundle branch block significantly increases risk of pacemaker...
Quentin Fischer (Department of Cardiology, Quebec Heart and Lung Institute, Laval University, Quebec City, Canada) and others report in Circulation: Cardiovascular Interventions that patients...
First US clinical study to assess ablation for monomorphic ventricular ablation...
Abbott has announced the start of LESS-VT—the first US clinical trial to evaluate the safety and effectiveness of ablation treatment for patients with monomorphic...
Tim Betts
For Tim Betts, a career in medicine was a natural choice. After graduating he knew he wanted to be a cardiologist and, with a...
AHA 2018: Treatment withdrawal should “not be attempted routinely” in patients...
Data from an open-label, pilot randomised trial (TRED-HF) indicate that if heart failure treatment is withdrawn in patients with recovered dilated cardiomyopathy, many will...
US FDA plan shakeup of its 510(k) clearance programme
The US FDA has announced plans to modernise its 510(k) clearance programme for approving medical devices for the US market. Data show that about...
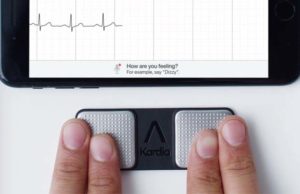
AHA 2018: Study shows AliveCor phone app can effectively identify STEMI...
An international study, led by researchers from the Intermountain Medical Center Heart Institute in Salt Lake City, found that a smartphone app (AliveCor) to...
Atrial fibrillation related to PFO closure occurs early and is transient
A new meta-analysis of patients undergoing percutaneous closure of patent foramen ovale (PFO) after cryptogenic shock supports previous studies that indicate PFO closure is...
Patients with new-onset atrial fibrillation after TAVI have highest risk of...
Amit N Vora (Duke University Medical Center/the Duke Clinical Research Institute, Durham, USA) and others report in in JACC: Cardiovascular Interventions that patients who...
Simultaneous TAVI and left atrial appendage closure is feasible
Thomas S Gilhofer (University Heart Center, Department of Cardiology, University Hospital Zurich, Zurich, Switzerland) and others report in Structural Heart that a combined procedure...
ESC 2018: British Heart Foundation to provide £30m for innovation in...
At the 2018 European Society of Cardiology (ESC) Congress (Munich, Germany), British Heart Foundation (BHF) announced the “Big Beat Challenge”—an £30m award that seeks...
ESC 2018: Siemens Healthineers showcase Magnetom Sola Cardiovascular Edition
At the 2018 European Society of Cardiology (ESC) Congress (25–29 August, Munich, Germany), Siemens Healthineers launched Magnetom Sola Cardiovascular Edition—a 1.5 Tesla magnetic resonance...
European Society of Cardiology Congress: Cardioskin launched
WeHealth, by Servier, has launched Cardioskin—a connected solution that is designed to function as an ambulatory wearable 15-lead ECG—at the 2018 European Society of...
More than 100,000 people now know hands-only CPR thanks to AHA...
More than 100,000 people have been trained in hands-only cardiopulmonary resuscitation (CPR) since the American Heart Association (AHA) launched its hands-only CPR training kiosk...
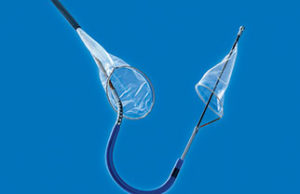
Novel Ultraseal device is a feasible option for left atrial appendage...
A feasibility study of a novel left atrial appendage device (Ultraseal, Cardia) indicates that the device is associated with a high rate of procedural...
Re-evaluate use of anticoagulation in patients with atrial flutter
A new study, published in JAMA Network Open, indicates that patients with atrial flutter have a lower incidence of stroke than patients with atrial...
Boston Scientific to buy cerebral protection system company
Boston Scientific has signed an agreement to acquire Claret Medical, which developed and commercialised the Sentinel cerebral embolic protection system. The device is used...
Funding for Neptune left ventricular assist device
A press release reports that the CALYPSO programme will receive 14 million euros to develop CorWave Neptune—a new type of cardiac support designed to...
Corwave announces issuance of US Patent Covering its implantable left ventricular...
CorWave has announced that it has obtained patent no. US 9,968,720 B2 on May 15, 2018 from the US Patent and Trademark Office. Titled...
One in five UK doctors say understaffing has affected patient safety
The annual census of UK consultants and higher speciality trainees—Focus on Physicians 2017–18—indicates that more than half of all consultants and two thirds of...
Cardiology societies come together to provide key advice for dealing with...
The American College of Cardiology (ACC), Heart Rhythm Society (HRS), North American Society for Cardiovascular Imaging (NASCI), Society for Cardiovascular Angiography and Interventions (SCAI),...
Janssen seeks approval for rivaroxaban to be used to reduce cardiac...
Janssen has submitted a supplemental new drug application to the US FDA for two new indications for rivaroxaban (Xarelto, which is marketed by Bayer in...
TCT 2017: Boston Scientific reveals presentations at conference
Boston Scientific has announced key data will be presented at next week’s Transcatheter Cardiovascular Therapeutics (TCT) meeting (29 October–2 November, Denver, USA). For example,...
Cardiology societies to provide new assessment options for subspecialities
The American College of Cardiology (ACC), Heart Failure Society of America (HFSA), Heart Rhythm Society (HRS) and Society for Cardiovascular Angiography and Interventions (SCAI)...
Pamela B Morris to be next American College of Cardiology scientific...
Pamela B Morris has been selected as the next vice chair of the American College of Cardiology (ACC) annual scientific session. She will serve...
American College of Cardiology says that reforms to US healthcare come...
The president of the American College of Cardiology (ACC), Mary Norine Walsh, has said that “it is concerning” that the Graham-Cassidy proposal (for US...
Three studies support efficacy of PFO closure for reducing risk of...
The results of CLOSE, Gore Reduce and the extended follow-up of RESPECT—all published in The New England Journal of Medicine—indicate that percutaneous closure of...
Familial screening after sudden death in young patients is effective even...
Pauline Quenin (I’institut du thorax, INSERM, CNRS, UNIV Nantes, CHU Nantes, Nantes, France) and others report in Circulation Arrhythmia and Electrophysiology that a quarter...
Data confirm HeartLogic Diagnostic helps to categorise patients at low or...
Boston Scientific has announced new data from the MultiSENSE (Multisensor chronic evaluation in ambulatory heart failure patients) study, which is evaluating the performance of...
RE-CIRCUIT data to be included in European summary of product characteristics...
The committee for medicinal products for human use (CHMP) of the European Medicines Agency (EMA) has issued a positive opinion for an update to...
Comprehensive consensus document on lead management published
The Heart Rhythm Society (HRS) has issued an international consensus statement that comprehensively addresses lead management for patients with cardiovascular implantable electronic devices (CIEDs).
The...
FDA grants 510(k) clearance to Fysicon’s QMAPP haemodynamic monitoring system
Fysicon has announced that it has received 510(k) FDA clearance for its QMAPP haemodynamic monitoring system. A press release reports that QMAPP offers cardiologists...
Kevin Campbell becomes new chief executive officer of PaceMate
PaceMate has announced the appointment of Kevin Campbell as its new chief executive officer (CEO). A press release reports that Campbell’s work as a...
Significant differences among atrial fibrillation patients across Asia
The latest results from one of the largest, ongoing global disease registries in atrial fibrillation (AF)— GARFIELD-AF (global anticoagulant registry in the field-atrial fibrillation)...
AtriClip Pro•V device launched onto US market
AtriCure has launched its AtriClip Pro•V left atrial appendage exclusion system in the USA. A press release reports that the new device offers an...
Caucasians have a higher incidence of an out-of-hospital cardiac arrest but...
There is a lack of sufficient data about the effect of racial differences on outcomes following an out-of-hospital cardiac arrest. Therefore, Karuppiah Arunachalam and...
ESC 2017: New research confirms significance of AliveCor’s 30-second ECG
AliveCor, the leader in FDA-cleared personal ECG technology, have announced the results of four clinical research presentations that demonstrate that AliveCor's hyper-fast, 30 second,...
ESC 2017: New subanalysis highlights benefits of edoxaban over warfarin in...
New subanalysis data demonstrate edoxaban (Lixana, Daiichi Sankyo) provide comparable efficacy and greater safety compared to warfarin, across non-valvular atrial fibrillation patients with different...
ESC 2017: Atrial fibrillation associated with a high financial, economic, and...
The first health economics data from GARFIELD-AF (Global anticoagulant registry in the field–atrial fibrillation) was presented at the European Society of Cardiology (ESC) congress...
ESC 2017: Rivaroxaban significantly lowers risk of stroke, cardiovascular death, and...
The COMPASS study, which was presented at the European Society of Cardiology (ESC) congress (26–30 August, Barcelona, Spain), has shown that rivaroxaban (Xarelto, Bayer)...
ESC 2017: EHRA and Biotronik mark 10 years of collaboration with...
At the European Society of Cardiology (ESC) congress (26–30 August, Barcelona, Spain), the European Heart Rhythm Association (EHRA) released an analytical supplement to the...
ESC 2017: Apixaban lowers risk of stroke in atrial fibrillation patients...
Data from the EMANATE trial, which was presented at a Hot Line session of the European Society of Cardiology (ESC) congress (26–30 August, Barcelona,...
Abbott’s Full MagLev HeartMate 3 LVAD receives FDA approval
The FDA has approved the Full MagLev HeartMate 3 (Abbott) left ventricular assist device for use in heart failure patients in need of short-term...
US trial to evaluate safety and efficacy of Amplatzer for patent...
Abbott has initiated a US pivotal clinical study evaluating the safety and effectiveness of a modified version of its Amplatzer device to correct patent...
ESC 2017: Case builds for dropping aspirin from triple therapy in...
The RE-DUAL PCI trial indicates that dual therapy with the non-vitamin K antagonist oral anticoagulant dabigatran (Pradaxa, Boehringer Ingelheim)—and not aspirin—and a P2Y12 inhibitor...
CSI 2017: Global postmarket surveillance study to explore use of LAmbre...
LifeTech, at the 2017 Congenital and Structural Intervention Congress (CSI; 28 June—1 July, Frankfurt, Germany), announced the launch of a three-year global postmarket surveillance...
Philips to acquire Spectranetics “to accelerate expansion” in its image-guided therapy...
Philips and Spectranetics have announced that they have entered into a definitive merger agreement. Pursuant to the agreement, a press release reports, Philips will...
ESC calls for sex-specific drug dosages of cardiovascular medication
Sex-specific cardiovascular drug dosages are needed to reduce adverse reactions in women, according to a position paper from the European Society of Cardiology (ESC)....
AHA 2016: HeartMate 3 LVAS improves clinical outcomes compared with HeartMate...
A prespecified primary analysis of the MOMENTUM 3 US IDE clinical study has indicated improvement in clinical outcomes for advanced heart failure patients implanted...
Patient enrolment complete in study assessing use of dabigatran atrial fibrillation...
Patient enrolment into the international Phase IIIb RE-DUAL PCI study is complete. The study is evaluating the safety and efficacy of dabigatran (Pradaxa, Boehringer...
Amulet study shows high implant success rates with Amplatzer Amulet LAA...
A study presented at the last late-breaking trial session of the 2016 Transcatheter Cardiovascular Therapeutics (TCT) meeting (29 October-2 November, Washington, DC, USA) indicates...
World’s smallest single-lead, medical grade ECG launched
AliveCor has announced the availability of Kardia Band—the world’s smallest single lead medical grade ECG. Kardia Band works with Apple Watch to provide a...
First patient procedure completed in UNCOVER-AF study
Acutus Medical has announced the completion of the first patient procedure in the UNCOVER-AF (Utilising novel dipole density capabilities to objectively visualise the etiology...
FDA clears temporary pacing lead for use during TAVI and other...
BioTrace Medical has received FDA 510(k) clearance for its Tempo Lead, which a press release describes as an innovative temporary pacing lead that has...
New vice president of US market development for CVRx
CVRx has appointed Tom Moore to the newly created, officer position of vice president, US Market Development. In this role, Moore will lead all field efforts...
MultiPoint™ pacing CRT technology shows promise in improving heart failure responder...
A growing body of evidence, including the most recent randomised clinical trial results presented at HRS and CARDIOSTIM-EHRA EUROPACE, has shown that cardiac resynchronisation therapy...