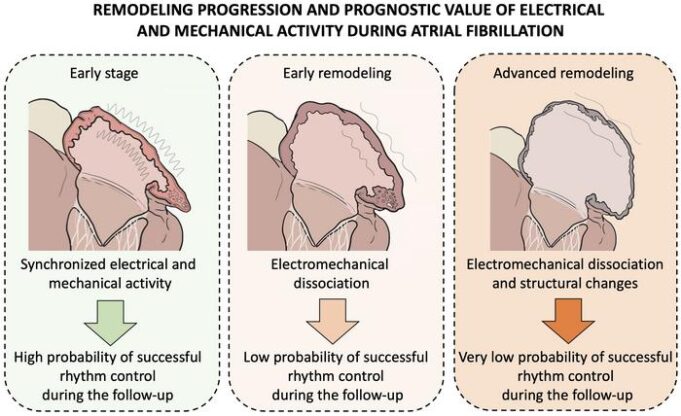
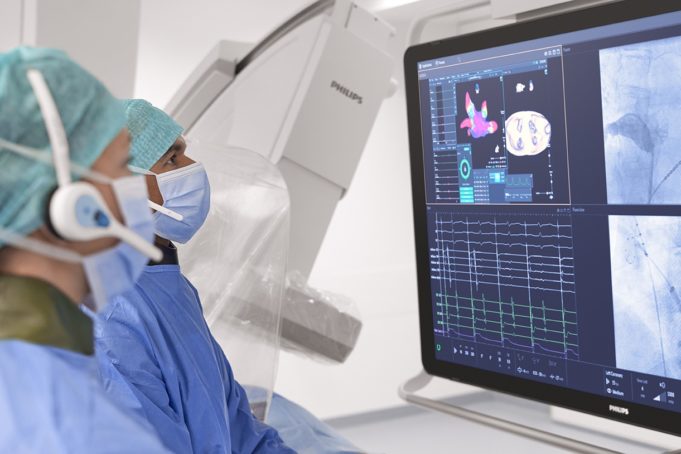
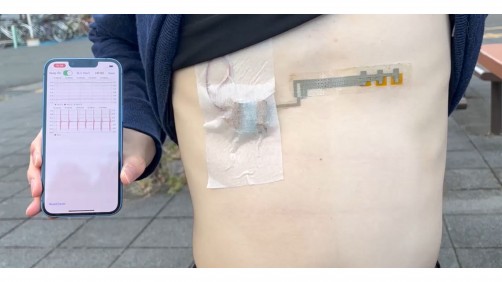
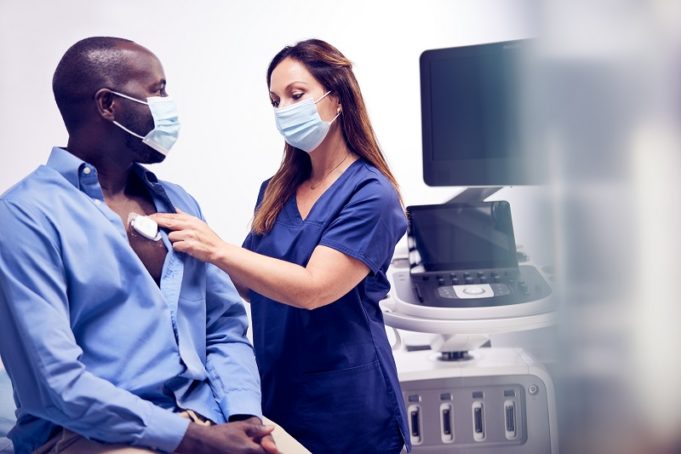
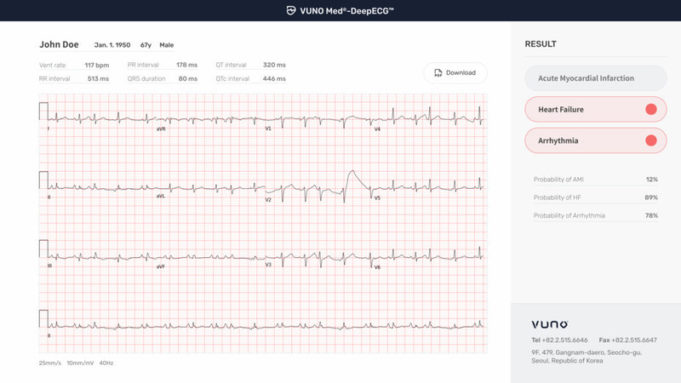
A new study analysing data from more than 13,000 patients has found that people using the increasingly popular weight-loss drugs known as glucagon-like peptide-1 receptor agonists (GLP-1-RAs) have a reduced risk of atrial fibrillation (AF), even after accounting for...
A new expert consensus statement published recently in the journal Heart Rhythm details the latest recommendations aimed at guiding clinicians and patients in choosing the right cardiovascular implantable electronic device (CIED), and managing these devices over time to reduce...
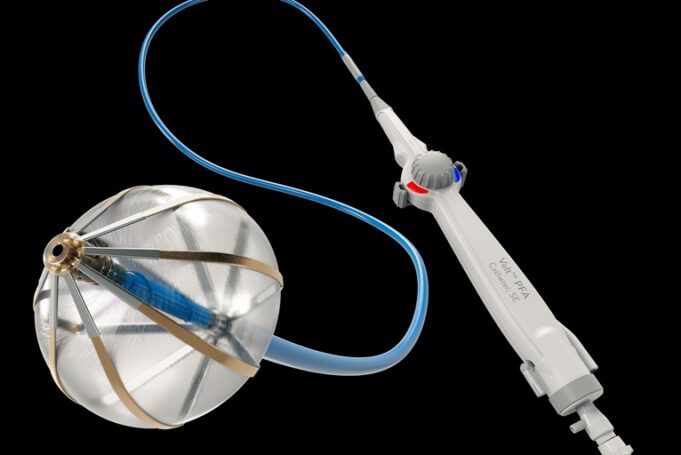
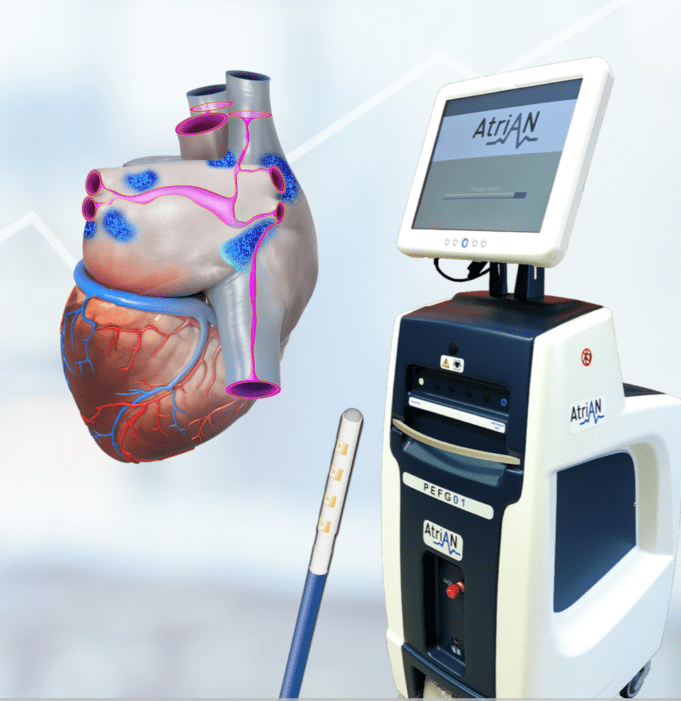
Abbott has announced late-breaking data from four trials that demonstrate strong clinical outcomes within its pulsed field ablation (PFA) and conduction system pacing (CSP) portfolios to treat heart rhythm disorders.
The data—presented at the 2026 Heart Rhythm Society (HRS) annual...
Johnson & Johnson (J&J) has announced the initiation of the PERSIGMA randomised controlled trial (RCT)—one of the first randomised studies evaluating competitive pulsed field ablation (PFA) technologies. The company claims that, in this rapidly advancing field, there is an...
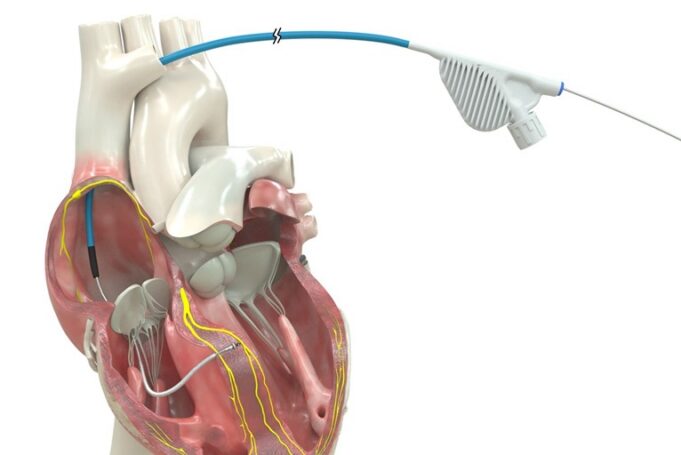
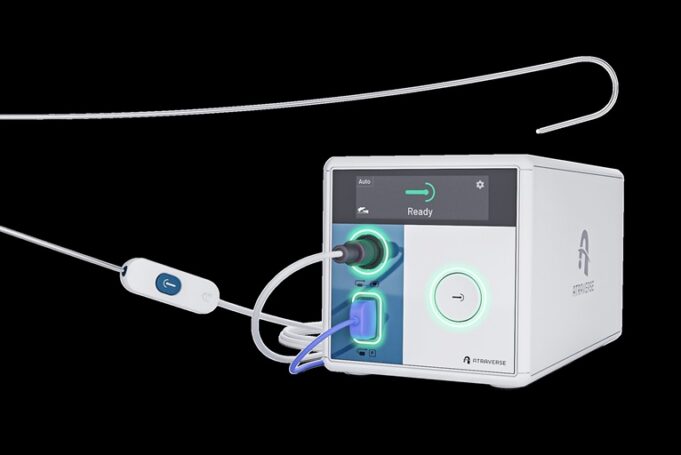
Atraverse Medical has announced accelerating market uptake for its Hotwire transseptal access system driven by rapid adoption in catheter labs nationwide and expanding use among electrophysiologists across the USA, according to a company press release.
The release adds that this...
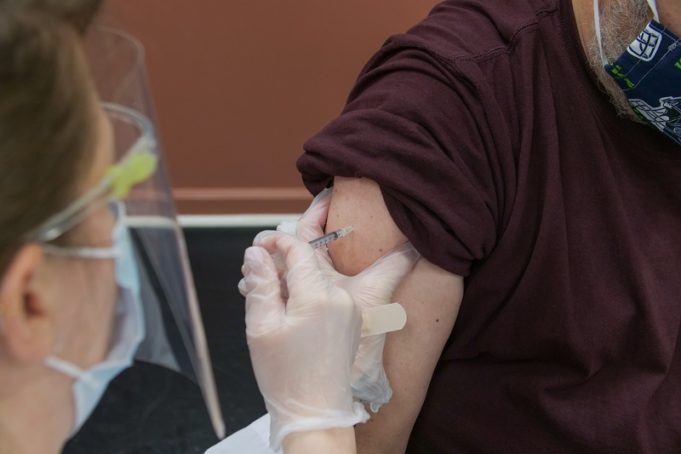
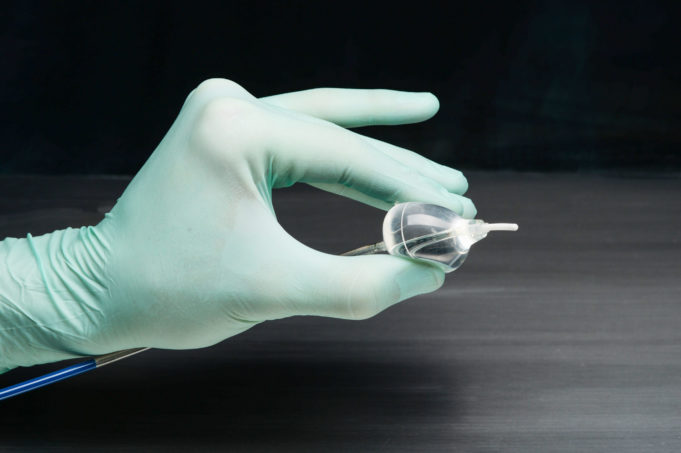
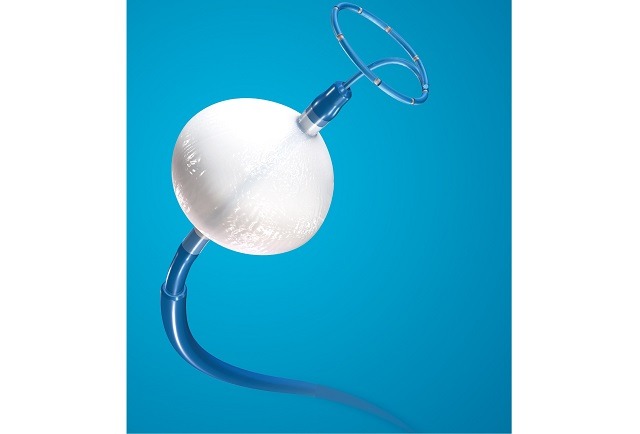
Boston Scientific Corporation has announced data supporting the use of the company's Farapulse pulsed field ablation (PFA) platform and Watchman left atrial appendage closure (LAAC) technologies. All data were presented at the 2026 Heart Rhythm Society (HRS) annual meeting...
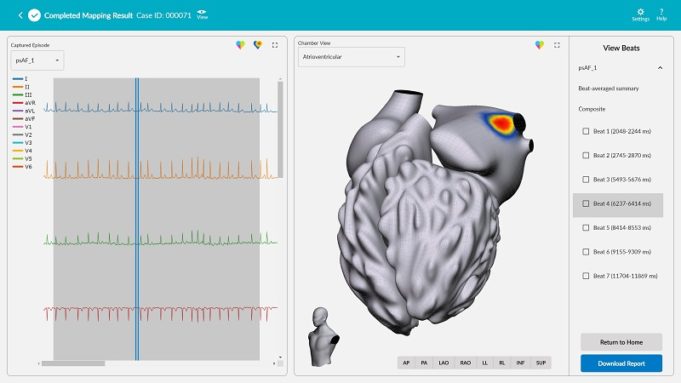
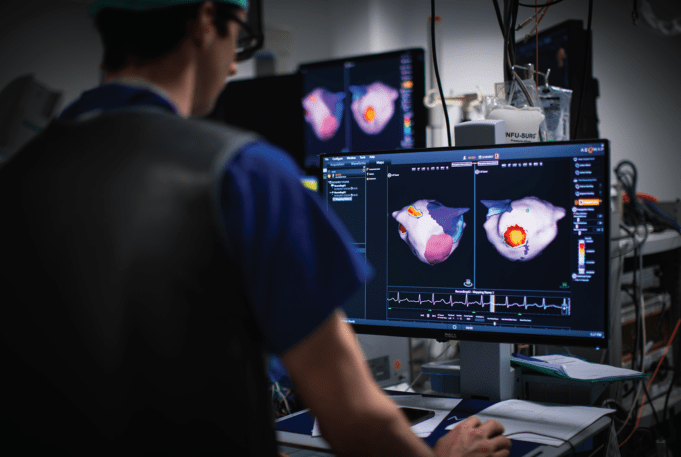
Corify Care has announced a “major clinical milestone” with the publication of two studies in the journals Heart Rhythm and EP Europace validating its proprietary technology as a “highly accurate solution” for mapping both atrial and ventricular arrhythmias in...
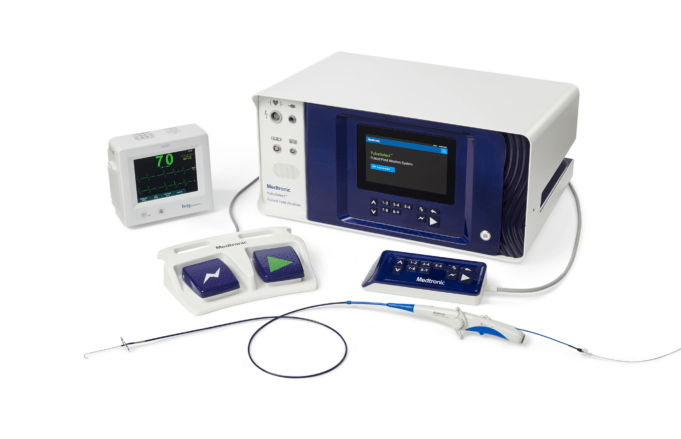
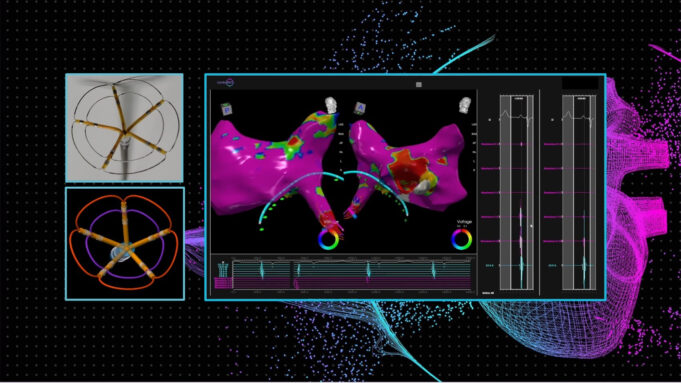
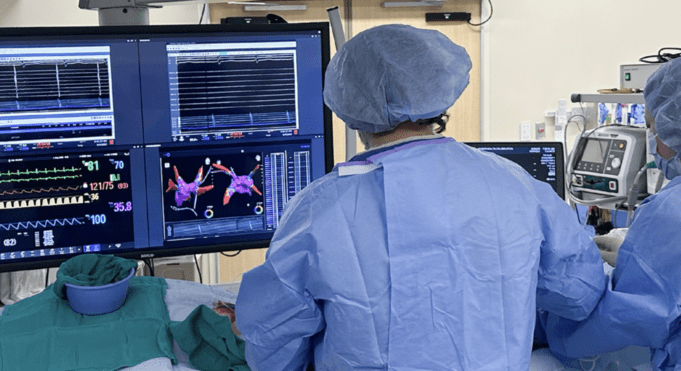
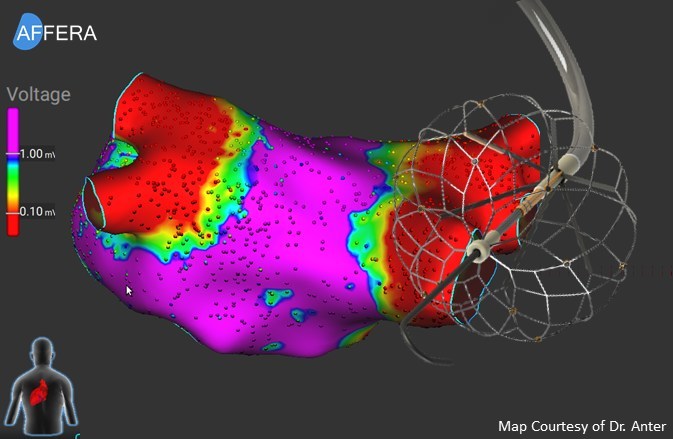
Medtronic has announced continued momentum regarding its Affera family of technologies for cardiac arrhythmia treatment, including promising data presented at the Heart Rhythm Society (HRS) annual meeting (23–26 April, Chicago, USA) and the start of a new trial to...
Haemonetics Corporation has announced the publication of a new study comparing its Vascade MVP XL vascular closure system with the Vascade MVP venous vascular closure system in the context of large‐bore venous access closure procedures. The study was published...
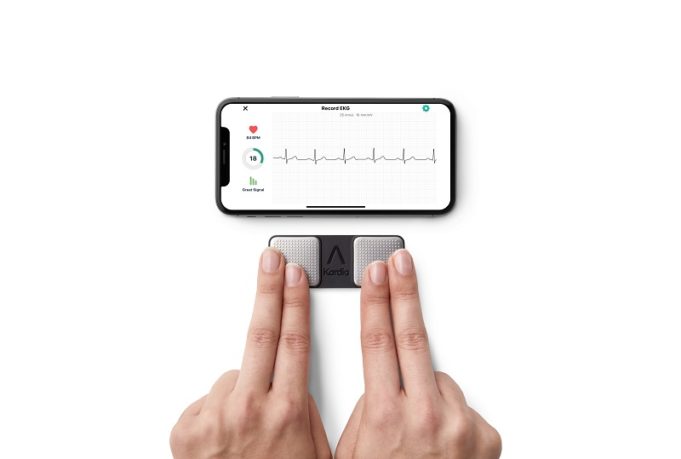
AliveCor has announced it has received a CE mark for the Kardia 12L electrocardiogram (ECG) system powered by the company’s KAI 12L artificial intelligence (AI) technology. Kardia 12L is described by the company as the “world's first” AI-powered, portable...
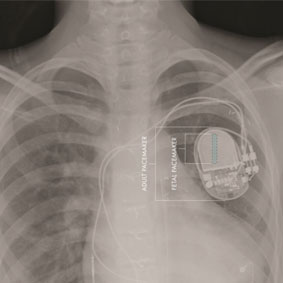
Biotronik has announced in a press release that—via a late-breaking trial session at the annual congress of the European Heart Rhythm Association (EHRA; 12–14 April 2026, Paris, France)—principal investigator Valentina Kutyifa (University of Rochester, Rochester, USA) recently presented new...
Initial one-year outcomes from the Leadless ARRIVE real-world study—presented at the annual congress of the European Heart Rhythm Association (EHRA; 12–14 April 2026, Paris, France) and simultaneously published in EP Europace—have demonstrated that Abbott’s Aveir AR leadless atrial pacemaker significantly...
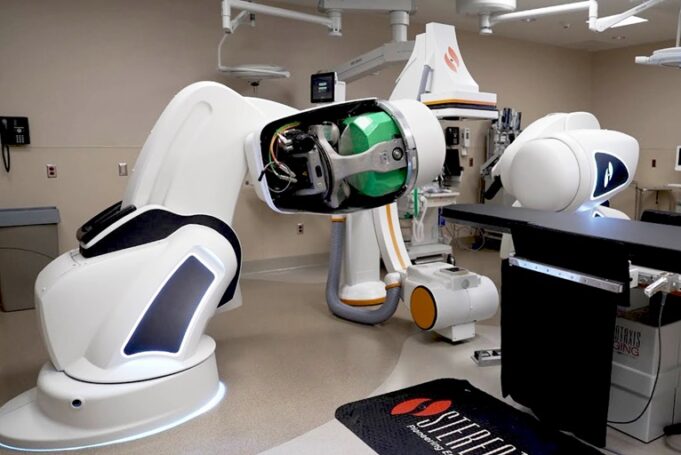
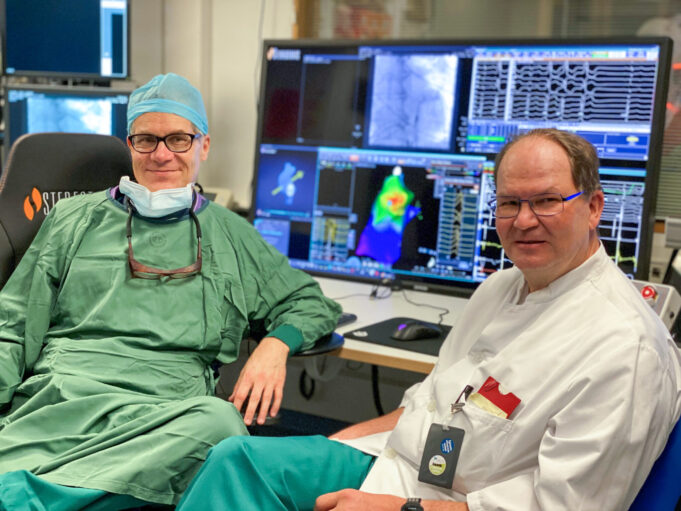
Stereotaxis has entered into a definitive agreement to acquire Robocath, a venture-backed innovator of robotic technologies for interventional cardiology and neurointerventions.
“Robocath represents a highly strategic addition to Stereotaxis, amplifying and accelerating our strategy as the leading robotic platform for...
A late-breaking presentation at the annual congress of the European Heart Rhythm Association (EHRA; 12–14 April 2026, Paris, France) has reaffirmed the safety and effectiveness of Boston Scientific’s Farapulse pulsed field ablation (PFA) platform, according to the company.
Within the BEAT...
Implicity has revealed new research presented at the annual congress of the European Heart Rhythm Association (EHRA; 12–14 April 2026, Paris, France) examining why false-positive alerts remain a persistent challenge for implantable cardiac monitors (ICMs)—even in devices equipped with...
Johnson & Johnson (J&J) has announced favourable 12-month interim effectiveness and safety results from VARIPURE—the largest prospective post-market follow-up study evaluating first-time pulsed field ablation (PFA) using the company’s Varipulse platform. These data were presented at the PFA Summit during...
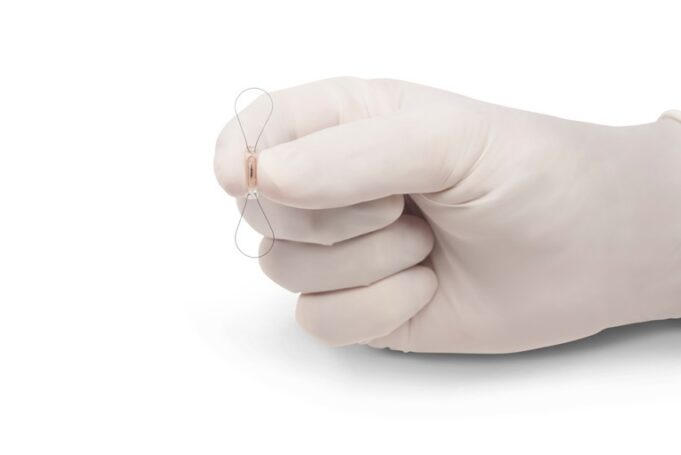
Protaryx Medical has received 510(k) clearance from the US Food and Drug Administration (FDA) to market its proprietary transseptal puncture device.
According to the company, this milestone represents a significant advancement in its mission to redefine safe, efficient and reproducible...
The US Food and Drug Administration (FDA) has reminded more than 2,200 medical product companies and researchers of requirements for them to submit certain information on clinical trial results to ClinicalTrials.gov.
As stated in a press release from the US...
Left bundle branch area pacing (LBBAP) has been found to be an effective and practical strategy for cardiac resynchronisation therapy (CRT), according to late-breaking science from the LECART trial presented today at the annual congress of the European Heart...
Heart failure is common in people who have atrial fibrillation (AF) detected during screening, according to a presentation delivered today at the annual congress of the European Heart Rhythm Association (EHRA; 12–14 April 2026, Paris, France).
“Heart failure and AF...
Haemonetics Corporation has announced US Food and Drug Administration (FDA) approval to expand the labelling for the Vascade MVP XL venous vascular closure system to include procedures using 10–14Fr inner diameter (ID) and up to 17Fr outer diameter (OD)...
Johnson & Johnson (J&J) has announced the launch of Varipulse Pro in Europe following CE-mark approval, advancing its pulsed field ablation (PFA) portfolio with a new pulse sequence that marks the “next evolution” of the company’s Varipulse platform, and...
PaceMate has announced the appointment of Benjamin A Steinberg as its chief medical officer (CMO).
Described by the company as a nationally recognised leader in cardiac electrophysiology, US National Institutes of Health (NIH)-funded researcher, and author of nearly 200 peer-reviewed...
Stereotaxis has announced that it has received US Food and Drug Administration (FDA) 510(k) clearance for the Synchrony system, which is designed to “digitise and modernise” the interventional cath lab.
“We have long recognised that seamless remote connectivity and collaboration...
Omron Healthcare has announced the launch of a randomised controlled trial in collaboration with the University of California San Francisco (UCSF; San Francisco, USA) to evaluate a new model for the early detection of atrial fibrillation (AF) in hypertension...
Boston Scientific Corporation announced today that it has received CE-mark approval for its Site Selective Pacing Catheters (SSPC) NXT.
These catheters are designed to support precise and controlled lead placement in the left bundle branch area (LBBA) and other right ventricular (RV) pacing...
The global community of heart rhythm specialists will unite later this month at the annual congress of the European Heart Rhythm Association (EHRA; 12–14 April 2026, Paris, France) with the goal of “shining a light” on cardiac arrhythmia management....
Results from the multicentre CRT‑NEXT study have shown that CRT‑DX (Biotronik)—a two-lead cardiac resynchronisation therapy defibrillator (CRT-D) system—is as safe and effective as conventional three-lead CRT‑D therapy. As per a Biotronik press release, the study’s findings, which were presented...
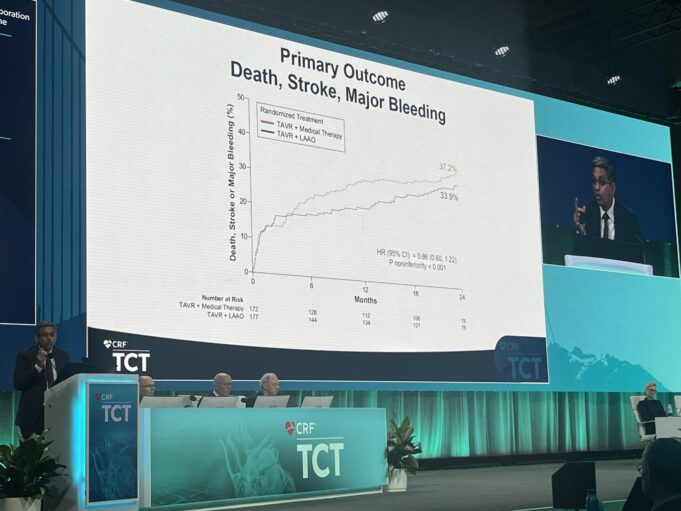
For patients with non-valvular atrial fibrillation (AF) who had no contraindications to taking oral anticoagulants, the use of a device to close off the left atrial appendage (LAA) in the heart was comparable to standard medication therapy in reducing...
Biotronik has announced the initiation of a global pivotal study evaluating its LivIQ leadless pacemaker system—a single‑device solution designed to deliver atrioventricular (AV) synchrony using advanced electrical far‑field sensing.
The first implantations within this study have been completed at the...
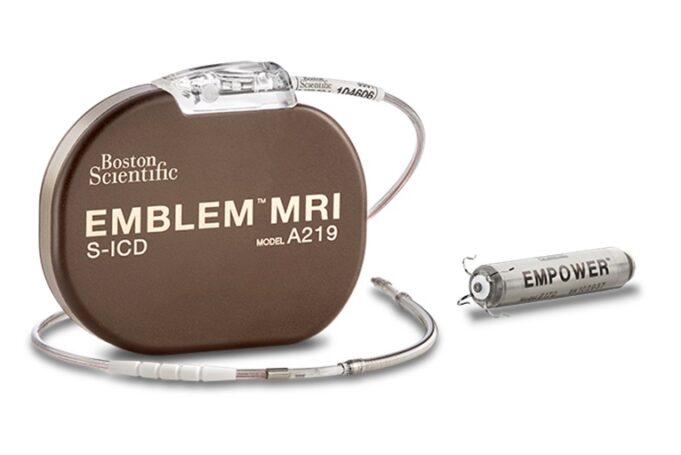
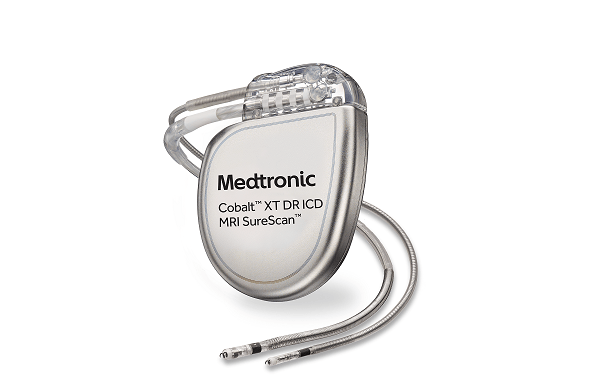
Medtronic has received US Food and Drug Administration (FDA) approval for an expanded indication with its OmniaSecure defibrillation lead. Now approved for placement in the left bundle branch (LBB) area, the lead can be used for conduction system pacing...
Luma Vision has announced the expansion of its clinical programme with the first patient enrolments in two additional European studies at Na Homolce Hospital with Vivek Reddy (Mount Sinai, New York, USA) and Petr Neuzil (Na Homolce Hospital, Prague,...
Nine medical societies are calling for updated safety standards in fluoroscopy laboratories, where clinicians face radiation exposure and orthopaedic injuries from heavy protective equipment.
A report published simultaneously today in the Journal of the Society for Cardiovascular Angiography and Interventions (JSCAI), Heart Rhythm, Journal of...
Johnson & Johnson (J&J) has announced the arrival of a new pulsed field ablation (PFA) technology for the treatment of drug-refractory paroxysmal atrial fibrillation (AF) in Peru, representing a “significant advance” in cardiac arrhythmia care with benefits including safety,...
Field Medical has announced the appointment of Mark Turco as chief executive officer (CEO), effective immediately.
As per a Field Medical press release, Turco brings more than 30 years of leadership experience spanning venture-backed cardiovascular startups and executive roles within...
New research led by scientists from the University of Colorado (CU) Anschutz (Aurora, USA) suggests that staying physically active after heart rhythm treatment may significantly reduce the risk of atrial fibrillation (AF) returning, offering patients a simple, low-cost way...
Abbott announced recently that its Volt pulsed field ablation (PFA) system is being used in procedures across the UK to treat people with atrial fibrillation (AF), with 15 National Health Service (NHS) hospitals taking part in a pilot study that...
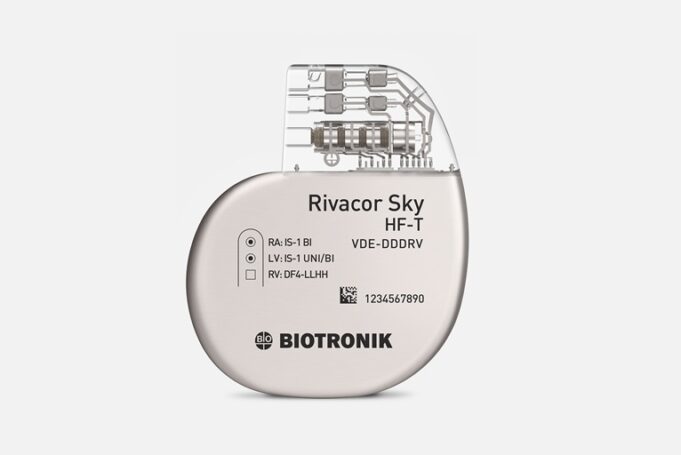
Biotronik has announced the launch of the new Acticor Sky and Rivacor Sky device family featuring “the world's first” CE mark-approved left bundle branch area pacing (LBBAP) high-voltage device, according to a press release from the company. These next-generation...
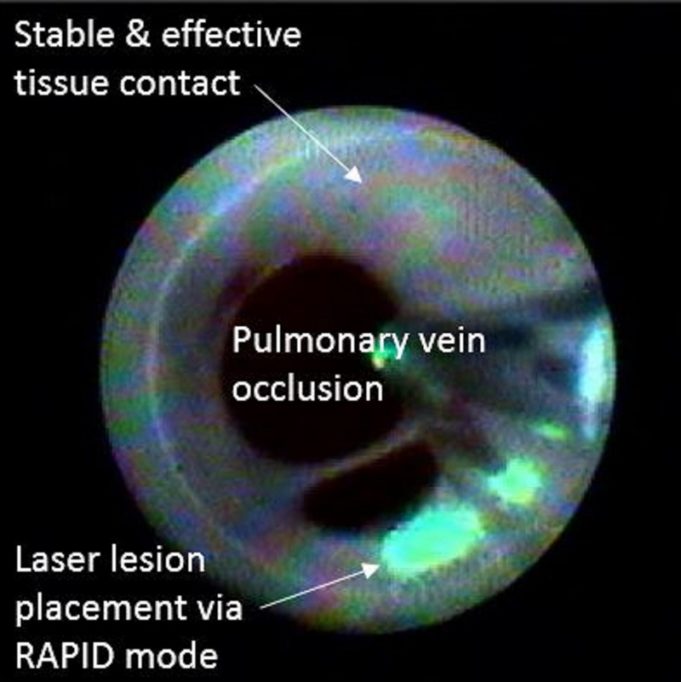
Pulse Biosciences has announced late-breaking clinical data from the nPulse cardiac catheter first-in-human feasibility study, demonstrating the successful treatment of atrial fibrillation (AF) across 150 patients alongside rapid procedure times and minimal adverse effects. The data were presented at...
Atraverse Medical has announced the presentation of new clinical and preclinical data at the 2026 AF symposium (5–7 February, Boston, USA), further validating the safety, performance and workflow advantages of the company’s Hotwire transseptal access system.
According to Atraverse, findings...
Abbott has announced new clinical data from two late-breaking presentations at the 2026 AF symposium (5–7 February, Boston, USA) that demonstrate the safety and efficacy of the company's minimally invasive therapies to treat people with atrial fibrillation (AF)—including 12-month findings...
Argá Medtech has announced positive clinical results from the BURST-AF first-in-human trial, demonstrating the safety, effectiveness and durability of its Coherent sine-burst electroporation (CSE) system for the treatment of paroxysmal and persistent atrial fibrillation (AF). Data presented at the...
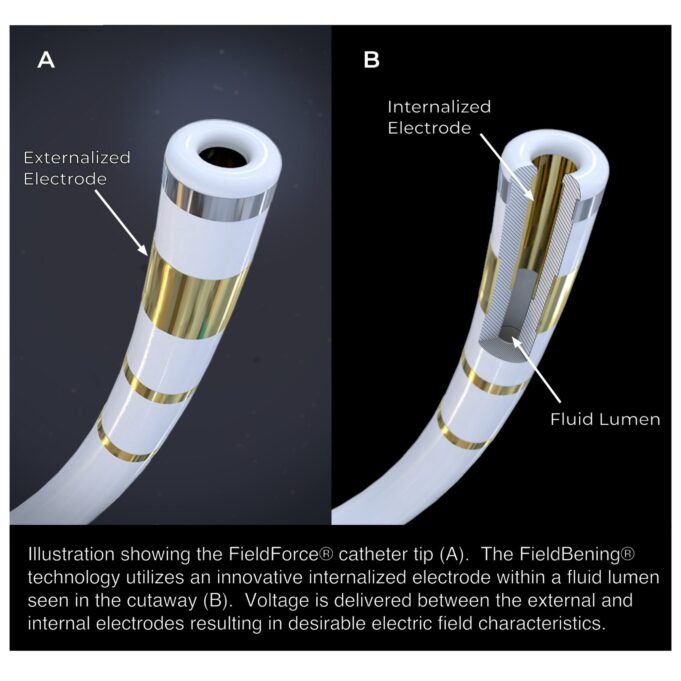
Field Medical has announced the presentation of late-breaking clinical trial data from the first-in-human Field PULSE study—which is evaluating the company’s FieldForce ablation system and catheter—at the 2026 AF symposium (5–7 February, Boston, USA).
Field PULSE is assessing a novel...
Johnson & Johnson (J&J) has announced 12-month pilot-phase data from the OMNY-AF study evaluating its investigational Omnypulse platform for the treatment of symptomatic paroxysmal atrial fibrillation (AF) during the 2026 AF symposium (5–7 February, Boston, USA). Initial outcomes across...
Volta Medical has announced results from the RESTART trial, published in the journal Heart Rhythm, that demonstrate positive outcomes for patients with recurrent atrial fibrillation (AF) and isolated pulmonary veins who underwent ablation using the company’s artificial intelligence (AI) solution...
The UK’s National Institute for Health and Care Excellence (NICE) has issued guidance recommending Abbott’s CardioMEMS HF system for remote monitoring of adults with chronic heart failure who have been hospitalised and are at risk of further admission.
The paperclip-sized...
An international research consortium has announced the launch of AF-B-STEP, a four-year research project designed to improve how atrial fibrillation (AF) is detected, quantified and treated worldwide. AF-B-STEP brings together 18 partners from Europe and Canada with the collective...
Corify Care has announced a new publication on its proprietary Global Volumetric Mapping technology in Nature Communications Medicine. The company says this technology represents the first system capable of mapping all four heart chambers at once, providing physicians with a...
Cathvision has announced “significant clinical and scientific progress” in pulsed field ablation (PFA) with its PFAnalyzer software module, which the company says reinforces its differentiated position in electrophysiology (EP) recording and analysis. The work demonstrates how high-fidelity unipolar electrograms...
Biotronik has today announced the successful first-in-human implantations of its LivIQ leadless pacemaker system as part of the BIO|CONCEPT.LivIQ study—a premarket clinical investigation designed to evaluate the system’s preliminary safety and performance.
The initial procedures were performed in Australia by Paul Gould at...
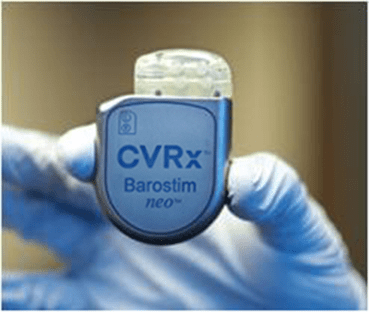
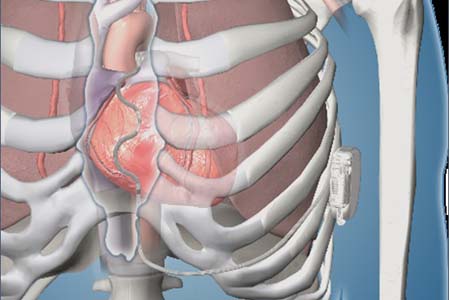
CVRx recently announced the start of the BENEFIT-HF clinical trial, a randomised study designed to evaluate the impact of the Barostim neuromodulation device in a "significantly expanded heart failure population", according to the company.
The trial is supported by Centers...
Medtronic has today announced two major milestones relating to the Affera Sphere-360 catheter—the receipt of a CE mark in Europe and completion of the first cases in the Horizon 360 investigational device exemption (IDE) pivotal trial in the USA.
“The Sphere-360 catheter offers...
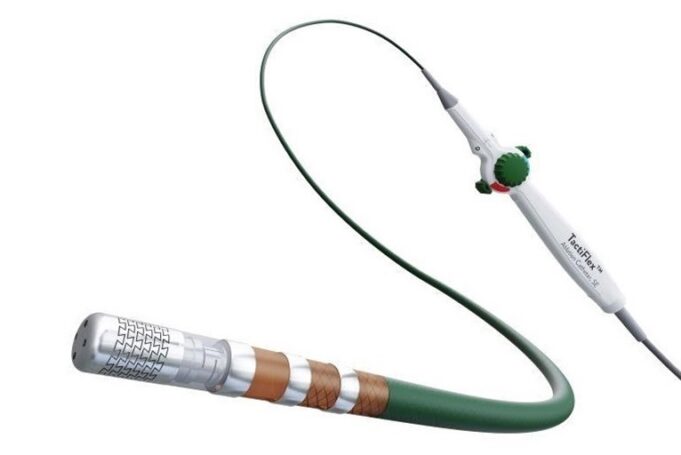
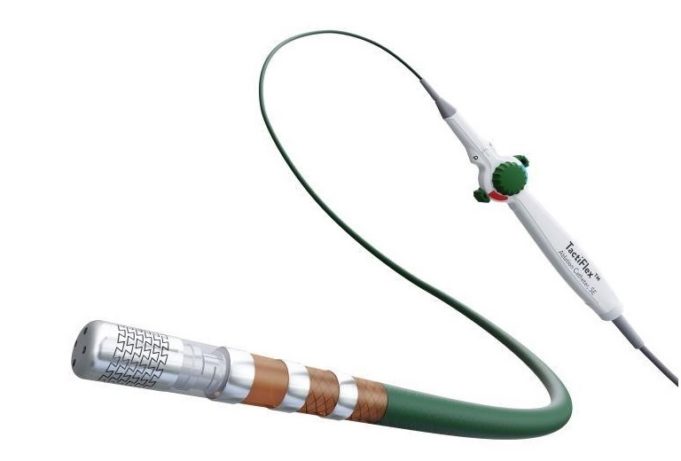
Abbott announced this week that it has received a CE mark in Europe for the sensor-enabled TactiFlex Duo ablation catheter to treat patients with atrial fibrillation (AF). Following this new approval, the first successful commercial cases using TactiFlex Duo...
1. Use of antidepressant medication linked to substantial increase in sudden cardiac death risk
The use of antidepressant (AD) medications has been linked to a substantial increase in the risk of sudden cardiac death (SCD), as per research presented at the...
Medtronic has commercially launched the OmniaSecure defibrillation lead in the USA this week, with the first cases being performed at hospitals across the country.
OmniaSecure is described by the company as “the world’s smallest defibrillation lead”. It has been approved...
AliveCor has announced it has received US Food and Drug Administration (FDA) clearance for the next generation of KAI 12L—the artificial intelligence (AI) technology powering its Kardia 12L electrocardiogram (ECG) system—to detect five additional cardiac determinations, bringing the total...
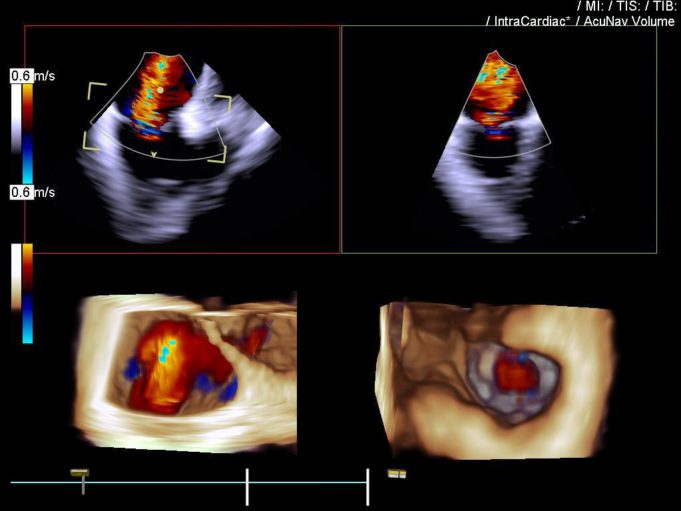
YorLabs has announced the successful completion of the first clinical procedure using its US Food and Drug Administration (FDA) 510(k)-cleared intracardiac imaging system.
The procedure marks the first real-world clinical use of YorLabs’ Zero-CapEx imaging platform following US FDA clearance....
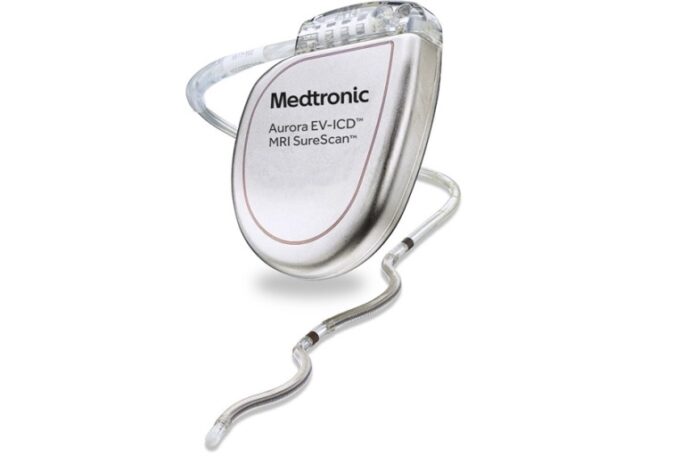
Abbott has announced a collaboration with AtaCor Medical to advance a next-generation investigational extravascular implantable cardioverter defibrillator (EV-ICD) system designed to deliver defibrillation therapy to people with life-threatening heart rhythms.
AtaCor is a cardiac rhythm management company developing extravascular defibrillation...
Accurkardia has announced US Food and Drug Administration (FDA) 510(k) clearance and the commercial launch of its AccurECG analysis system (v2.0). AccurECG 2.0 builds on AccurKardia’s previously US FDA-cleared AccurECG platform, representing the next-generation evolution of its "enterprise-grade" electrocardiogram...
Luma Vision has announced the successful completion of 15 Verafeye-guided persistent atrial fibrillation (AF) ablation procedures conducted as part of a clinical study in collaboration with Cardiofocus.
The study is designed, in part, to evaluate the ability of Luma Vision's...
Stereotaxis has today announced that it has obtained US Food and Drug Administration (FDA) approval for the Magic magnetic interventional ablation catheter—a robotically navigated catheter designed to perform cardiac ablation procedures for the treatment of heart arrhythmias.
“US FDA...
WL Gore & Associates—Gore—has announced that it has entered into a definitive agreement to acquire Conformal Medical, developer of the CLAAS AcuFORM system, a left atrial appendage occlusion (LAAO) technology. The transaction is expected to close in early 2026,...
Abbott has announced that the US Food and Drug Administration (FDA) has approved the company's Volt pulsed field ablation (PFA) system to treat patients with atrial fibrillation (AF). Abbott says it will soon begin commercial PFA cases in the...
Cathvision has announced the appointment of Eric Thepaut as the company’s independent chairman of the board, and the commercial release of version 3.5 of its ECGenius system.
Eric Thepaut joins Cathvision following his recent retirement from Boston Scientific Corporation, where...
HeartBeam has announced that the US Food and Drug Administration (FDA) has granted 510(k) clearance for the company’s “groundbreaking” 12-lead electrocardiogram (ECG) synthesis software for the assessment of arrhythmias. This clearance follows HeartBeam’s successful appeal of a prior Not...
Boston Scientific Corporation has received a CE mark for the Farapoint pulsed field ablation (PFA) catheter, which is intended for use in the treatment of right atrial flutter (AFL) by administering ablation to the cavotricuspid isthmus (CTI) area of...
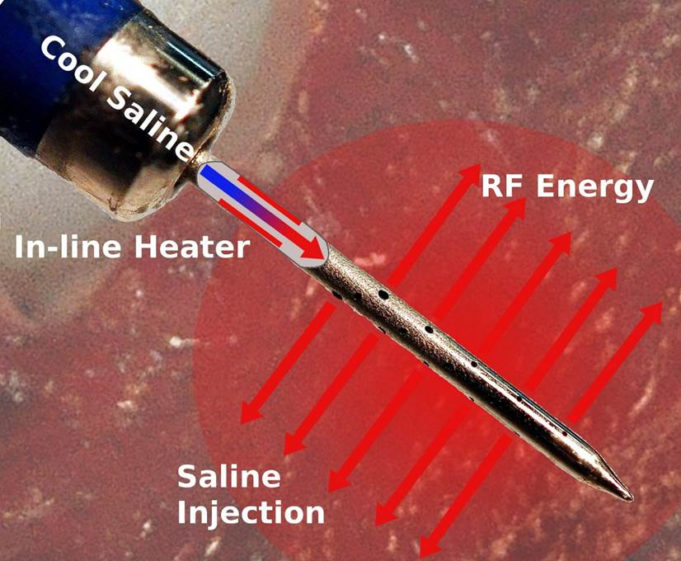
Atraverse Medical has received 510(k) clearance from the US Food and Drug Administration (FDA) for its fully integrated Hotwire transseptal access system.
The novel system includes the Hotwire radiofrequency (RF) generator featuring impedance-guided shutoff after transseptal crossing—mitigating the risk of...
VisCardia has secured US$40 million in growth financing to advance the company’s path toward premarket approval (PMA) for its VisONE synchronised diaphragmatic stimulation (SDS) therapy.
The financing will support the execution of the company’s pivotal RECOVER-HF clinical study, and key...
Medtronic has announced the recent presentation of results from two late-breaking studies at the 2025 Asia Pacific Heart Rhythm Society (APHRS) Scientific Sessions (12–15 November, Yokohama, Japan), including six-month results from the Enlighten study—a post-approval registry of the company’s...
Catheter ablation—a minimally invasive procedure to correct irregular heart rhythms—may reduce the risk of stroke to such an extent that some patients can discontinue blood thinners, according to preliminary late-breaking science presented at the 2025 American Heart Association (AHA)...
Biotronik has announced the enrolment of the first patient in the RESET-AF 3D study, a multicentre clinical study evaluating the Elepulse pulsed field ablation (PFA) system for its performance in the treatment of atrial fibrillation (AF) patients. The study...
For older people with irregular heart rhythms who are at high risk of stroke and bleeding, standard care—including the timely use of anticoagulant blood thinners, when indicated—has been found to be the better choice compared to a catheter-based procedure,...
People with atrial fibrillation (AF) and obesity may have fewer episodes of AF after catheter ablation treatment if they take the diabetes medication metformin in addition to standard care, according to preliminary, late-breaking science presented at the 2025 American...
New research has found that a simplified regimen of clot-preventing medication following stent placement in adults with atrial fibrillation (AF) was just as safe and effective in preventing strokes, heart attack and death when compared to a standard yearlong...
Volta Medical has announced the US launch of the AF-Xplorer II system, the company's next-generation artificial intelligence (AI) solution designed to simplify real-time assessments of complex atrial fibrillation (AF).
A Volta press release states that, building on the success of...
Johnson & Johnson (J&J) MedTech has announced that new clinical and real-world data from its integrated-by-design Varipulse platform in pulsed field ablation (PFA) procedures for atrial fibrillation (AF) will be presented at the Asia Pacific Heart Rhythm Society (APHRS) and...
An artificial intelligence (AI) algorithm paired with the single-lead electrocardiogram (ECG) sensors on a smartwatch accurately diagnosed structural heart diseases, a study to be presented at the 2025 American Heart Association scientific sessions (7–10 November, New Orleans, USA) has...
AtriCure has announced that the first patient was recently enrolled and treated in the BoxX-NoAF clinical trial, which is assessing the safety and effectiveness of box lesion creation with the company's Isolator Synergy EnCompass clamp and AtriClip left atrial...
YorLabs, a medical technology company developing next-generation intracardiac imaging solutions for electrophysiology (EP) and interventional cardiology (IC) procedures, has announced that the US Food and Drug Administration (FDA) has granted 510(k) clearance for its YorLabs intracardiac imaging system—a “first-of-its-kind”,...
Stereotaxis and CardioFocus have announced that they have entered into a collaboration agreement to advance robotic pulsed field ablation (PFA) technology towards commercialisation.
This collaboration is set to combine the proprietary technologies of both companies to accelerate commercial availability of the “first...
A prospective study indicates that the prevalence of new-onset atrial fibrillation (AF) after coronary artery bypass graft (CABG) surgery may be significantly higher than previously reported, but the extremely low burden of the disease calls into question the need...
Conformal Medical has announced the initiation of patient enrolment in the European Union (EU) for the CONFORM pivotal trial. The first EU procedure was successfully performed at the Institut Cardiovasculaire Paris Sud (ICPS) in Paris, France by Philippe Garot.
The...
Biotronik has today announced the successful first-in-human implantation of a device from its new Acticor/Rivacor Sky implantable cardioverter defibrillator (ICD)/cardiac resynchronisation therapy-defibrillator (CRT-D) portfolio that has been specifically engineered to support conduction system pacing via left bundle branch area...
Omron Healthcare has announced that cumulative worldwide sales of Omron blood pressure monitors (BPMs) have exceeded 400 million units.
This underscores the company’s longstanding commitment to extending healthy life expectancy by advancing home blood pressure monitoring and preventive cardiovascular care,...
Stereotaxis has announced the successful first procedures by physicians at Erasmus University Medical Center in Rotterdam, The Netherlands, using the company’s Genesis robotic magnetic navigation system.
“We have long recognised the clinical benefits of robotics, and are delighted to be...
Boston Scientific has received CE-mark approval to expand the indication for its current-generation Ingevity+ pacing leads—thin wires that are implanted in the heart and connected to a pacemaker. This expansion now includes conduction system pacing (CSP) and sensing of...
Argá Medtech has announced initial enrolments in the COHERENT-AF pivotal clinical trial. This US Food and Drug Administration (FDA) investigational device exemption (IDE), prospective, non-randomised trial is designed to evaluate the safety and effectiveness of the Coherent Sine-Burst Electroporation...
Medtronic has announced the initiation of a pivotal study evaluating the use of elevated and personalised cardiac pacing rates for the treatment of patients with heart failure with preserved ejection fraction (HFpEF).
While physicians routinely use pacing therapy to...
AccurKardia has announced the initiation of a multicentre clinical pilot study of AK+ Guard—a US Food and Drug Administration (FDA) Breakthrough Device designated, artificial intelligence (AI)-powered technology that enables the detection of hyperkalaemia using Lead I electrocardiograms (ECGs). The...
An individually tailored, multidimensional risk-based treatment strategy was not associated with improvements in clinical outcomes compared with usual guideline-based care in patients with atrial fibrillation (AF), according to late-breaking research presented at the 2025 European Society of Cardiology (ESC)...
Biotronik has announced the market release of Solia CSP S—the latest innovation in its growing portfolio of conduction system pacing (CSP) solutions. Solia CSP S is the “first and only” pacing lead worldwide that combines a fixed screw design with a stylet-driven implantation...
HeartBeam has announced new study data demonstrating no significant differences in the detection of atrial fibrillation (AF), atrial flutter and sinus rhythm when the company’s deep learning algorithms were applied to the HeartBeam system, or to standard 12-lead electrocardiograms...
At the 2025 European Society of Cardiology (ESC) congress (29 August–1 September, Madrid, Spain), Boston Scientific shared two late-breaking presentations pertaining to its Farapulse pulsed field ablation (PFA) system and Watchman FLX left atrial appendage closure (LAAC) device.
New findings...
Procedure-related infection rates are similar between reconditioned and new pacemakers, potentially offering a "new hope" to patients in low- and middle-income countries (LMICs), according to late-breaking research presented at this year’s European Society of Cardiology (ESC) congress (29 August–1...
In recognition of National Atrial Fibrillation (AF) Awareness Month in the USA (September), StopAfib.org has launched the 2025 ‘Get in Rhythm. Stay in Rhythm.’ AF awareness campaign.
StopAfib.org notes in a recent press release that it designated September as AF...
Adding digital twin-guided ablation to a standard ablation technique can improve outcomes in patients with persistent atrial fibrillation (AF), according to late-breaking research presented at the 2025 European Society of Cardiology (ESC) congress (29 August–1 September, Madrid, Spain).
“Ablation using...
Kardium has announced that it has received premarket approval (PMA) for the Globe pulsed field ablation (PFA) system, and 510(k) clearance for both the Globe introducer sheath and the Globe PFA system mapping software, from the US Food and...
Pulsed field ablation (PFA) did not demonstrate superior efficacy to radiofrequency (RF) ablation in patients with drug-resistant, paroxysmal atrial fibrillation (AF) within the BEAT-PAROX-AF trial, from which late-breaking findings were presented at the European Society of Cardiology (ESC) congress...
Laza Medical has announced the successful completion of its first software-only human cases at a US medical institution, marking the first time its real-time, artificial intelligence (AI)-based heart model has been evaluated alongside real clinical procedures.
The software operated concurrently...
Stereotaxis has announced the successful completion of the world’s first procedures using Magic Sweep—the “first and only” robotically navigated, high-density electrophysiology (EP) mapping catheter. The procedures were performed by Raffaele Corbisiero and Pedram Kazemian at Deborah Heart and Lung...
Researchers recently presented three new datasets reinforcing the safety and performance of Medtronic’s OmniaSecure defibrillation lead at the 2025 European Society of Cardiology (ESC) congress (29 August–1 September, Madrid, Spain), as per a press release from the company.
The global...
Prophylactic implantable cardioverter defibrillator (ICD) therapy does not appear to reduce mortality in patients with a prior myocardial infarction (MI), persistent moderate left ventricular (LV) systolic dysfunction and abnormal electrocardiogram (ECG) markers, according to late-breaking research presented at the...
Johnson & Johnson (J&J) MedTech has today announced acute safety and effectiveness results from the VARIPURE substudy of the SECURE study evaluating the company’s Varipulse platform in pulsed field ablation (PFA) procedures for atrial fibrillation (AF).
The real-world data—presented at...
A mail-based atrial fibrillation (AF) screening programme with electrocardiogram (ECG) patch monitoring led to a modest long-term increase in AF diagnosis and anticoagulation exposure in older patients at moderate-to-high stroke risk within a late-breaking trial presented at the European...
Discontinuing oral anticoagulation (OAC) therapy may result in a lower risk of a composite of stroke, systemic embolism or major bleeding versus continuing OAC therapy in patients who have undergone successful ablation for atrial fibrillation (AF) at least 12...
Targeting high-normal potassium levels can reduce the risk of arrhythmias, hospitalisation for heart failure or arrhythmia, and death, compared with no intervention, according to late-breaking research presented at the 2025 European Society of Cardiology (ESC) congress (29 August–1 September,...
Conformal Medical has announced the successful closing of its Series D extension financing, with a new partner joining inside investors to raise a total of US$32 million.
The funds will support enrolment in the company's ongoing CONFORM pivotal trial and...
Orchestra BioMed has announced the publication of data in the Journal of the American College of Cardiology: Clinical Electrophysiology from a pressure-volume (PV) loop study demonstrating favourable hemodynamic effects of atrioventricular interval modulation (AVIM) therapy in pacemaker-indicated patients with...
Pulsecare Medical has announced that its innovative NxPFA nanosecond pulsed field ablation (ns-PFA) system has received marketing approval from China's National Medical Products Administration (NMPA).
As the “world's first” third-generation ns-PFA system utilising high-voltage nanosecond pulses for pulmonary vein isolation (PVI) in...
Omron Healthcare announced recently that it has successfully concluded a three-month study demonstrating how remotely monitored vital signs can facilitate early medical intervention in patients with heart failure.
Conducted in collaboration with Kyoto Prefectural University of Medicine (Kyoto, Japan), with...
Royal Philips has announced a collaboration through which the company will integrate its suite of cardiac ambulatory monitoring and diagnostics services with Aura, the specialty diagnostics suite of leading electronic health record (EHR) company Epic.
According to Philips, this collaboration will...
Orchestra BioMed has secured US$70 million in new capital from Ligand Pharmaceuticals and Medtronic to advance its late-stage partnered cardiology programmes—arterioventricular interval modulation (AVIM) therapy and the Virtue Sirolimus AngioInfusion Balloon (Virtue SAB).
Simultaneously, Orchestra BioMed and Medtronic, which have...
Cardiosense has announced that the US Food and Drug Administration (FDA) has granted 510(k) clearance for the CardioTag device—claimed by the company to be the first multimodal, wearable sensor that simultaneously captures high-fidelity electrocardiogram (ECG), photoplethysmogram (PPG) and seismocardiogram...
Stereotaxis has announced that it has received US Food and Drug Administration (FDA) 510(k) clearance for its Magic Sweep catheter—described by the company as the “world’s first” robotically navigated high-density electrophysiology (EP) mapping catheter. Stereotaxis claims in a press...
Biotronik has announced a strategic distribution agreement with CardioFocus to advance ablation treatment for cardiac arrhythmias, with Biotronik serving as the exclusive distributor of the Centauri pulsed field ablation (PFA) system in selected European countries.
A Biotronik press release states that this...
DeepQure has announced that South Korea's Ministry of Food and Drug Safety (MFDS) has approved a clinical trial of the company’s HyperQure renal denervation (RDN) system—a novel laparoscopic RDN system for the treatment of atrial fibrillation (AF).
The trial is designed to...
Scar tissue in the heart—myocardial fibrosis—may be associated with abnormal heart rhythms among healthy, long-time male endurance athletes aged 50 years or older, potentially increasing their risk of sudden cardiac death, as per the findings of a small study...
Researchers from the University of California San Francisco (UCSF) in San Francisco, USA recently set out to better understand the relationship between transseptal puncture holes and visual auras as a result of catheter ablation, and found that small injuries...
AtriCure has announced the completion of enrolment in the LeAAPS clinical trial—a prospective, randomised, blinded, superiority investigational device exemption (IDE) trial evaluating the AtriClip left atrial appendage (LAA) exclusion system for the prevention of ischaemic stroke and systemic arterial...
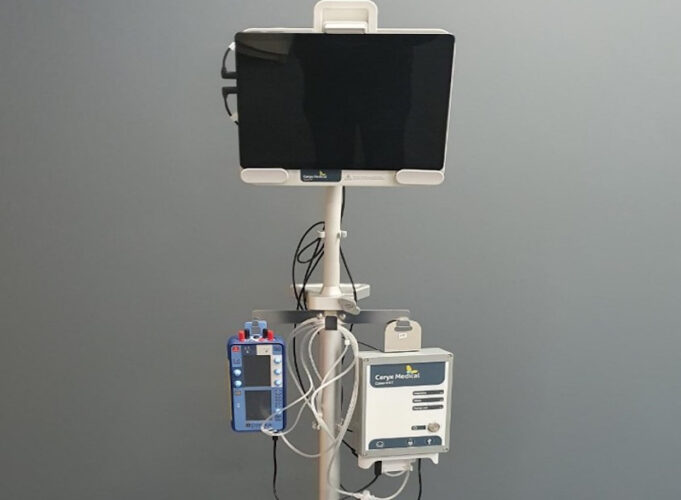
Ceryx Medical has announced the successful close of a new funding round, bringing the company’s total raised to US$15 million.
The company’s lead product—a temporary cardiac pacing device—is currently undergoing clinical evaluation. Early results from the ongoing study are “highly...
Royal Philips has announced the launch of the Philips electrocardiogram (ECG) artificial intelligence (AI) marketplace—a platform that gives cardiac care teams access to multiple vendor offerings all in one central location to help clinicians manage and implement AI-powered diagnostic tools...
Patients with heart failure and atrial fibrillation (AF) at the time of mitral transcatheter edge-to-edge repair (M-TEER) for severe mitral regurgitation (MR) are more than twice as likely to die or be rehospitalised for heart failure (HF), compared to...
Boston Scientific Corporation has received US Food and Drug Administration (FDA) approval to expand the instructions for use (IFU) labelling for the Farapulse pulsed field ablation (PFA) system. The updated labelling now includes approval for the system in the...
Stereotaxis has announced the publication in the Journal of Interventional Cardiac Electrophysiology of initial clinical results from a first-in-human study utilising the MAGiC catheter—the first peer-reviewed piece of literature supporting the efficacy and safety of the company’s robotically navigated,...
Field Medical and Kardium—two companies developing pulsed field ablation (PFA) systems for the treatment of heart rhythm disorders—have secured funding rounds worth US$35 million and US$250 million, respectively.
Earlier this month, Field Medical announced the close of an oversubscribed US$35 million Series...
Cathvision has announced the market release of ECGenius system version 3.4, introducing multiple software enhancements aimed at improving procedural workflow and physician usability across and between electrophysiology (EP) labs.
A press release from the company notes that specific features of...
Johnson & Johnson (J&J) MedTech has announced the launch of the Varipulse platform—a system used to perform catheter ablation procedures for the treatment of atrial fibrillation (AF)—in the Asia-Pacific region.
"PFA , as a new type of energy, has the potential...
The UK’s National Institute for Health and Care Excellence (NICE) has today published new guidance recommending the use of pulsed field ablation (PFA) in the treatment of atrial fibrillation (AF), citing the fact the procedure “works well” and there...
Merit Medical recently announced the appointment of Martha Aronson as the company's new president and chief executive officer (CEO), effective 3 October 2025. Fred P Lampropoulos will remain as chairman of the board, president and CEO of Merit through...
The presence of atrial fibrillation (AF) may reduce the likelihood of mechanical thrombectomy being successful at the first attempt in patients with acute ischaemic strokes caused by medium-vessel occlusions (MeVOs). That is according to a registry study published recently...
New research presented at the 2025 European Academy of Neurology (EAN) congress (21–24 June, Helsinki, Finland) has uncovered a notable link between nighttime heart rhythm and future health conditions—even in people with no obvious sleep problems.
The study, which was...
Vivasure Medical has announced the submission of a premarket approval (PMA) application to the US Food and Drug Administration (FDA) for its PerQseal Elite vascular closure system for arterial procedures.
The submission builds upon the successful results of the PATCH...
Volta Medical has announced a “significant update” to the US labelling of its flagship system, Volta AF-Xplorer. Labelling will now include “groundbreaking” clinical data from the TAILORED-AF trial, which showed significantly improved long-term outcomes compared to standard-of-care treatment for...
Ceryx Medical has announced the successful closure of a new funding round, bringing the company’s total raised to US$15 million, with the new investment intended to accelerate development of a permanent, physiologically responsive pacemaker designed to provide long-term support...
New regulations have come into effect in the UK which place a greater emphasis on medical device manufacturers to monitor the safety and performance of their products.
The new Post-Market Surveillance (PMS) regulations came into effect from Monday (16 June)...
The international STEEER-AF study has shown that targeted online education on atrial fibrillation (AF) for health professionals can improve guideline-adherent care. This cluster-randomised controlled trial, now published in Nature Medicine, was designed to test if structured online AF education for...
Atraverse Medical has announced the close of US$29.4 million in follow-on financing to accelerate growth, building upon the US$12.5 million in prior seed investment that was previously used to obtain US Food and Drug Administration (FDA) 510(k) clearance and...
Cardiac monitoring company PaceMate has announced the appointment of JR Finkelmeier as its new chief executive officer (CEO).
Finkelmeier joined PaceMate as chief commercial officer in 2024 after years leading Philips’ ambulatory monitoring division. Now, as CEO of PaceMate, he...
Medicare patients with atrial fibrillation (AF) who undergo surgical ablation during isolated coronary artery bypass grafting (CABG) live longer than those who do not, new research published in The Annals of Thoracic Surgery have shown.
Researchers examined Medicare claims data...
Johnson & Johnson (J&J) MedTech has announced the US launch of the Soundstar Crystal ultrasound catheter for intracardiac echocardiography (ICE) imaging in cardiac ablation procedures.
The device provides clearer, enhanced image quality compared to other ICE catheters, J&J MedTech claims, thus...
Omron Healthcare has introduced home blood pressure monitors with built-in, artificial intelligence (AI)-powered detection of atrial fibrillation (AF). In what the company describes as a “medical device first”, blood pressure monitors featuring Omron’s novel machine learning Intellisense AFib algorithm...
Royal Philips has today announced the introduction of its VeriSight Pro 3D intracardiac echocardiography (ICE) catheter in Europe. Building on its success in the USA, VeriSight Pro brings real-time 3D imaging directly inside the heart, helping physicians perform procedures with...
An artificial intelligence (AI)-enabled, electrocardiogram (ECG)-based algorithm has performed well in the early detection of heart failure among healthcare-seeking individuals in Kenya, according to late-breaking research presented today at Heart Failure 2025 (17–20 May, Belgrade, Serbia), a scientific congress...
This advertorial is sponsored by Boston Scientific.
Given the ever-increasing wealth of clinical data and experiences involving pulsed field ablation (PFA) for the treatment of atrial fibrillation (AF), purpose-built technologies like the FARAPULSETM PFA System (Boston Scientific) are currently garnering...
HeartBeam has announced that its “groundbreaking” synthesised 12-lead electrocardiogram (ECG) successfully met the clinical endpoints in the VALID-ECG pivotal study. Thomas Deering (Piedmont Heart Institute, Atlanta, USA) presented these data during the recent Heart Rhythm Society (HRS) annual meeting...
The TRANSFORM-AF study has found that patients with atrial fibrillation (AF) and obesity who were treated with a glucagon-like peptide-1 receptor agonist (GLP-1 RA) experienced a reduction in AF-related events—including hospitalisation, cardioversion and ablation for AF. This multicentre, pharmacoepidemiologic...
Circa Scientific has announced that the US Food and Drug Administration (FDA) has granted 510(k) clearance for the PeriCross epicardial access kit (formerly Rook), a purpose-built solution designed to enable controlled and efficient access to the pericardial space via...
Implicity has revealed new findings from its EVIDENCE-RM study, which—by leveraging extensive data from the French National Health Data System (SNDS)—has demonstrated clear clinical and medico-economic benefits associated with the company’s universal monitoring system (UMS) compared to conventional, manufacturer-specific...
New research unveiled at the 2025 Heart Rhythm Society (HRS) annual meeting (24–27 April, San Diego, USA) has underscored the critical role of artificial intelligence (AI) in improving the procedural safety of cardiac electrophysiology (EP), demonstrating how intelligent, real-time...
Among a great number of presentations relating to pulsed field ablation (PFA) treatments for atrial fibrillation (AF) at this year’s Heart Rhythm Society (HRS) annual meeting (24–27 April, San Diego, USA), industry leaders Abbott and Medtronic both announced the...
Kardium has announced late-breaking, one-year clinical trial results with its innovative pulsed field ablation (PFA) device—the Globe PF system.
Pivotal findings from the PULSAR investigational device exemption (IDE) study were announced at the 2025 Heart Rhythm Society (HRS) annual...
The Heart Rhythm Society (HRS) and the American College of Cardiology (ACC) have released a joint scientific statement on new guiding principles for same-day discharge (SDD) in patients undergoing intracardiac catheter ablation procedures. This document draws on global clinical...
Field Medical has announced that its FieldForce ablation system was recently featured in a scientific presentation at the 2025 Heart Rhythm Society (HRS) annual meeting (24–27 April, San Diego, USA).
A late-breaking presentation of the VCAS trial on 27 April...
Abbott has announced late-breaking data from the AVEIR conduction system pacing (CSP) acute clinical feasibility study, demonstrating the safety and performance of the company’s investigational Aveir CSP leadless pacemaker technology. According to Abbott, the study is the “world's first” assessment...
Medtronic has received US Food and Drug Administration (FDA) approval of the OmniaSecure defibrillation lead for placement within the right ventricle. The lead—based on the SelectSecure Model 3830 pacing lead and delivered via catheter—builds on Medtronic’s portfolio of lead...
Atraverse Medical has announced that the latest clinical evidence on the efficacy and safety of its Hotwire left heart access system were presented at the 2025 Heart Rhythm Society (HRS) annual meeting (24–27 April, San Diego, USA).
Devi Nair (St Bernards Healthcare, Jonesboro,...
Boston Scientific has announced positive 12-month primary endpoint results from the second phase of the ADVANTAGE AF clinical trial evaluating the use of the Farapulse pulsed field ablation (PFA) system and adjunctive use of the Farapoint PFA catheter in...
Johnson & Johnson (J&J) MedTech has announced positive initial three-month results from the Omny-IRE study evaluating the investigational Omnypulse platform in patients with paroxysmal atrial fibrillation (AF). The data, unveiled as a late-breaking presentation at the 2025 Heart Rhythm...
SoloPace Incorporated has announced both US Food and Drug Administration (FDA) clearance and first-in-human use of its SoloPace control system for temporary pacing in transcatheter aortic valve implantation (TAVI) procedures.
With standardised workflows, the control system is engineered to improve TAVI...
Stereotaxis announced recently that it hosted a demonstration of the GenesisX robotic system at this year’s Heart Rhythm Society (HRS) meeting (24–27 April, San Diego, USA), marking the first live, public demonstration of the technology and offering HRS attendees...
EBR Systems has received US Food and Drug Administration (FDA) approval of the WiSE system—marking what the company describes as a significant leap forward in the treatment of heart failure.
Unlike conventional cardiac resynchronisation therapy (CRT), the WiSE system...
The US Heart Rhythm Society (HRS) has released a framework outlining criteria for establishing an atrial fibrillation (AF) centre of excellence (CoE) and key operational standards to provide multidisciplinary care for AF patients.
A recent HRS press release notes that...
Field Medical has announced the successful closing of US$40 million in Series A financing. The round includes US$20 million in new capital and the conversion of US$20 million in seed-round debt. Participants include several leading strategics, venture capital firms and family offices, with multiple seed...
Orchestra BioMed Holdings has announced that the US Food and Drug Administration (FDA) has granted breakthrough device designation for atrioventricular interval modulation (AVIM) therapy.
The breakthrough device designation is for an implantable system (i.e. a pacemaker) to deliver AVIM therapy...
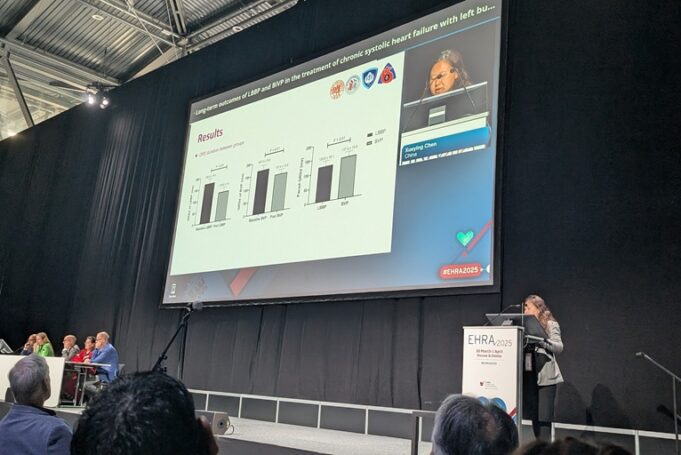
A left bundle branch pacing (LBBP) approach has been shown to be superior to biventricular pacing with regard to reducing long-term all-cause mortality and heart failure hospitalisation (HFH), according to new data showcased at the European Heart Rhythm Association...
Luma Vision has announced the US Food and Drug Administration (FDA) clearance of its Verafeye visualisation platform.
As per a company press release, this novel, catheter-based imaging system provides real-time, 2D/4D, 360-degree visualisation, “significantly enhancing” clinician precision and confidence...
Volta Medical—a company that develops artificial intelligence (AI) solutions to assist electrophysiologists—has announced the appointment of Bill Hoffman as executive chairman of its board of directors.
A press release states that Hoffman “brings a track record of guiding innovative startups to global...
AtriCure has announced the first use of the newest AtriClip device for minimally invasive concomitant procedures, the AtriClip PRO-Mini left atrial appendage (LAA) exclusion system.
The AtriClip PRO-Mini device received US Food and Drug Administration (FDA) 510(k) clearance earlier this...
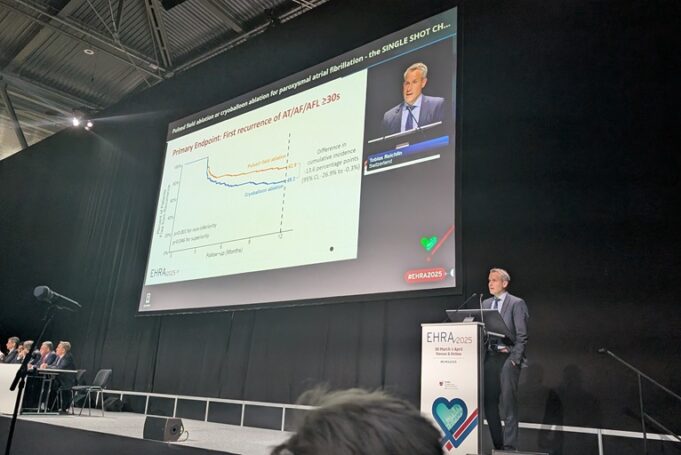
At this year’s European Heart Rhythm Association (EHRA) congress (30 March–1 April 2025, Vienna, Austria), a late-breaking presentation of data from the SINGLE SHOT CHAMPION study revealed the Farapulse pulsed field ablation (PFA) system (Boston Scientific) to be non-inferior—and...
The use of antidepressant (AD) medications has been linked to a substantial increase in the risk of sudden cardiac death (SCD), as per research presented at the recent European Heart Rhythm Association (EHRA) congress (30 March–1 April 2025, Vienna,...
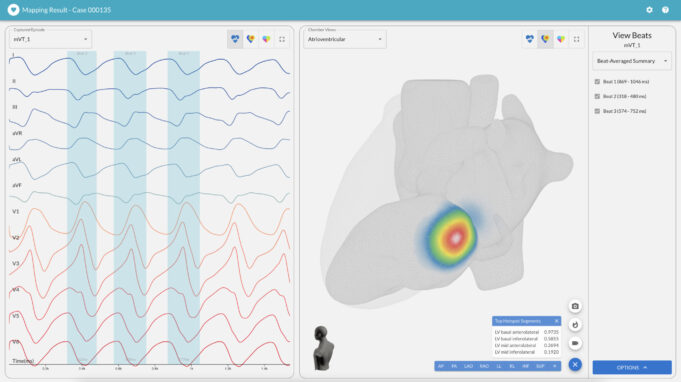
A novel cardiac ablation system may hold the potential to improve upon the currently available treatments for ventricular arrhythmia (VA) patients, having demonstrated positive safety signals in a European registry study including more than 100 of these typically challenging...
Pacemaker implantation within 30 days after a transcatheter aortic valve implantation (TAVI) procedure has been found to correlate with significantly higher rates of all-cause and cardiovascular-related mortality at up to 10 years of follow-up, as per data from the...
Medtronic recently announced that the first patient has been enrolled in a US Food and Drug Administration (FDA) early feasibility study evaluating the Affera mapping and ablation system with the Sphere-9 catheter using new investigational radiofrequency (RF) and pulsed...
A new study presented by researchers at the 2025 European Heart Rhythm Association (EHRA) congress (30 March–1 April, Vienna, Austria) demonstrates how, using artificial intelligence (AI) to analyse standard 12-lead electrocardiograph (ECG) data taken from almost half a million cases,...
Conformal Medical has announced the enrolment of its 500th patient in the CONFORM investigational device exemption (IDE) trial, the prospective, multicentre, randomised controlled study evaluating the safety and efficacy of the Conformal CLAAS AcuFORM system compared to other commercially...
The European Society of Cardiology (ESC) has released what it describes as a “groundbreaking” consensus statement on conduction system pacing (CSP), marking a “significant milestone in the evolution of pacing therapy”. The document was officially presented at the ongoing...
Johnson & Johnson (J&J) MedTech has today announced 12-month results from the SmartfIRE study, delivered as a late-breaking presentation at the ongoing European Heart Rhythm Association (EHRA) congress (30 March–1 April 2025, Vienna, Austria), with findings showing that use...
New findings from the SINGLE SHOT CHAMPION trial, presented at the ongoing European Heart Rhythm Association (EHRA) congress (30 March–1 April 2025, Vienna, Austria) by Tobias Reichlin (Bern University Hospital, Bern, Switzerland), have shown the Farapulse pulsed field ablation...
In a Cardiac Rhythm News guest article, chief investigators Nikolaos Dagres and Gerhard Hindricks (both Berlin, Germany) provide a window into the PROFID EHRA study—an international, investigator-initiated trial set up with the goal of redefining the prevention of sudden...
Abbott announced yesterday that it has received CE-mark approval in Europe for the Volt pulsed field ablation (PFA) system to treat patients with atrial fibrillation (AF). Following what the company describes as an “earlier-than-expected” CE mark, Abbott has begun...
This year’s congress of the European Heart Rhythm Association (EHRA; 30 March–1 April, Vienna, Austria), a branch of the European Society of Cardiology (ESC), is set to bring together experts in various disciplines—including sudden cardiac death, ventricular arrhythmias, device...
AtaCor Medical has announced that it has initiated its ASCEND EV study—a prospective, non-randomised international study aiming to evaluate the safety and performance of the company’s all-in-one extravascular implantable cardioverter defibrillator (EV-ICD) lead system in combination with leading commercially...
GE HealthCare has announced the launch of the AltiX AI.i edition of its Mac-Lab, CardioLab and ComboLab cath lab and electrophysiology (EP) lab systems.
The AltiX AI.i editions are designed to improve the user experience, elevating workflow in the cardiac...
A software robot has proved to be faster than doctors at detecting side-effects during a drug treatment for cardiac arrhythmia, while also cutting unnecessarily frequent follow-up lab tests and controls, according to a study conducted at the University of...
MedLumics has announced the publication of first-in-human results for the AblaView pulsed field ablation (PFA) system in atrial fibrillation (AF) patients, including clinical safety and the ability to predict durable lesions at three months with 100% specificity.
“We would...
Stereotaxis has announced a US Food and Drug Administration (FDA) regulatory submission for the first robotically navigated catheter designed to expand usage of robotic magnetic navigation into the broader endovascular field.
Emagin 5F is the first in a family of...
Cadrenal Therapeutics has announced the signing of a collaboration agreement with Abbott to support Cadrenal’s pivotal TECH-LVAD trial.
Cadrenal Therapeutics is a late-stage biopharmaceutical company focused on developing specialised therapeutics for rare cardiovascular conditions.
The company is developing its late-stage asset,...
DeepQure has announced progress of clinical trials of its extravascular renal denervation device, HyperQure, and is expanding its technology to address atrial fibrillation (AF), one of the most common cardiac arrhythmias.
The company is actively conducting clinical trials in both...
With cases of cardiac arrhythmias rising rapidly, cardiologists from the European Heart Rhythm Association (EHRA) have marked Pulse Day (1 March) by urging communities and healthcare systems to ‘Feel the Pulse’, and take simple steps to raise awareness of...
Boston Scientific’s vice president (VP) of rhythm management, Caroline Bravo, speaks to Cardiac Rhythm News about the arrival of pulsed field ablation (PFA) for atrial fibrillation, and her expectations for the technology in the coming year.
How much of a...
Research from Amsterdam UMC (Amsterdam, The Netherlands) and Johns Hopkins University (Baltimore, USA), published recently in the European Heart Journal, could represent an important step in the hunt for a ‘one-off’ gene therapy that can improve heart function and protect...
Following a temporary, voluntary pause of US external evaluation and all US cases with its Varipulse catheter, Johnson & Johnson (J&J) is set to resume the limited market release of these devices in the USA in light of a...
Cathvision has announced the receipt of a CE mark for its breakthrough product, the ECGenius system. According to the company, this regulatory milestone allows Cathvision to market and distribute its ECGenius system across the European Economic Area (EEA), bringing...
New findings from the DiVERT Stroke clinical study show that women spent less time in the hospital, saw fewer referrals to cardiology and were prescribed less frequent post-stroke cardiac monitoring than men when admitted to comprehensive stroke centres (CSCs)...
Boston Scientific Corporation has announced it has received CE-mark approval for its navigation-enabled Farawave Nav ablation catheter in the treatment of paroxysmal atrial fibrillation (AF) and for its new Faraview software. Both technologies will integrate and expand mapping option...
Volta Medical has announced the publication in Nature Medicine of the TAILORED-AF clinical trial, which demonstrated that an artificial intelligence (AI)-guided procedure for persistent atrial fibrillation (AF) in combination with conventional pulmonary vein isolation (PVI) treatment resulted in better outcomes...
First-in-human results of a novel transseptal puncture device from Protaryx Medical were presented at the 2025 Society of Thoracic Surgeons (STS) annual meeting (25–27 January, Los Angeles, USA).
The device is a novel catheter system designed to improve the safety,...
Stereotaxis has announced that it recently received European CE-mark approval for the MAGiC ablation catheter. A press release highlights this approval is a “significant milestone” for Stereotaxis and for the community of physicians pioneering robotics in electrophysiology—and one that...
Lucia and the Heart Rhythm Society (HRS) have announced a strategic collaboration to leverage Lucia's cutting-edge technology to enhance UpBeat.org—the HRS’ premier online resource for patients and caregivers. The atrial fibrillation (AF)-focused artificial intelligence (AI) enhancements will be pilot...
From first-time clinical data to major industry debuts, here are Cardiac Rhythm News’ 10 most popular stories of 2024.
1. Abbott joins the pulsed field ablation party
Early on in the year, Abbott announced the first global procedures had been conducted...
Researchers from Mass General Brigham (Boston, USA) have evaluated a drug that represents a new class of anticoagulants known as Factor XI inhibitors for treating patients with atrial fibrillation (AF) as part of the AZALEA-TIMI 71 study—reporting in the New England Journal...
Pulse Biosciences has announced late-breaking data from the first-in-human feasibility study of its nanosecond pulsed field ablation (nsPFA) 360-degree cardiac catheter system at the recent 30th annual AF symposium (16–18 January 2025, Boston, USA).
The feasibility study in question is intended...
Taho Pharmaceuticals has announced the completion of the pivotal trial for TAH3311, an anticoagulant oral dissolving film (ODF).
The trial was conducted following detailed discussions and alignment with the US Food and Drug Administration (FDA) and European Medicines Agency (EMA),...
AliveCor has announced that the US Centers for Medicare & Medicaid Services (CMS) has included the company's artificial intelligence (AI)-powered electrocardiogram (ECG) technology in the 2025 Hospital Outpatient Prospective Payment System (OPPS) final rule, assigning it to Ambulatory Payment...
Clinical evidence supporting the efficacy, safety and efficiency of Atraverse Medical's Hotwire left-heart access system was presented recently at the 30th annual AF symposium (16–18 January 2025, Boston, USA), the company has announced.
Atraverse states in a press release that...
Boston Scientific announced data supporting the use of the Farapulse pulsed field ablation (PFA) system and the Watchman FLX left atrial appendage closure (LAAC) device, respectively, during a late-breaking science session at the 30th annual AF symposium (16–18 January 2025, Boston,...
Luma Vision has announced the successful first-in-human device use and enrolment in its LUMINIZE clinical study involving the “groundbreaking” Verafeye platform. A company press release states that this cutting-edge, four-dimensional catheter-based imaging system is designed to “transform” the field of...
Primary results from the DEFINE AFib clinical study have found that Medtronic's LINQ family of insertable cardiac monitors (ICMs), paired with a novel algorithm, were able to detect atrial fibrillation (AF) episodes and properly risk stratify patients as high...
AliveCor has announced new data published in the journal Computing in Cardiology (CINC) demonstrating that its Kardia 12L electrocardiogram (ECG) system—the “world's first” artificial intelligence (AI)-powered, handheld 12-lead ECG system with a unique single-cable design—performs comparably to a standard, hospital-grade ECG...
Circa Scientific has announced the launch of its innovative Crosswise radiofrequency (RF) transseptal access system, which is designed to provide precise puncture of the septal wall with a streamlined, zero-exchange workflow.
The Crosswise system delivers RF energy directly to the tip...
Biotronik has announced the first in-human implantations in the BIO-Master.CSP study examining the use of the company’s investigational, next-generation Solia conduction system pacing (CSP) S pacing lead when implanted in the left bundle branch area (LBBA).
LBBA pacing is an...
Peerbridge Health has announced the launch of its COR-INSIGHT trial, which will validate the screening and diagnostic capabilities of its Peerbridge Cor ambulatory electrocardiography (ECG) wearables. The trial seeks to “revolutionise” cardiovascular and cardiopulmonary monitoring and screening by providing...
Johnson & Johnson MedTech has announced European CE mark approval of the Dual Energy Thermocool Smarttouch SF catheter for the treatment of cardiac arrhythmias.
The Dual Energy Thermocool Smarttouch SF catheter is an irrigated, contact-force sensing catheter powered by the...
The US Food and Drug Administration (FDA) has issued draft guidance that includes recommendations to support development and marketing of safe and effective artificial intelligence (AI)-enabled devices.
The guidance, if finalised, would be the first guidance to provide comprehensive...
Abbott has announced the successful completion of the “world's first” in-human leadless left bundle branch area pacing (LBBAP) procedures using the company's investigational Aveir conduction system pacing (CSP) leadless pacemaker system, as part of a feasibility study.
These procedures mark...
HeartBeam has announced that the US Food and Drug Administration (FDA) has granted 510(k) clearance of the HeartBeam system for comprehensive arrhythmia assessment. A press release from the company details that, with its patented design, the HeartBeam device is...
The US Food and Drug Administration (FDA) has alerted patients, caregivers and healthcare providers about the potential need for early device replacement of Boston Scientific’s Accolade pacemakers—including the Accolade, Proponent, Essentio and Altrua 2 Standard Life (SL) and Extended...
Field Medical has announced that its FieldForce ablation system has been accepted into the US Food and Drug Administration (FDA)’s Total Product Life Cycle Advisory Program (TAP) pilot and been granted breakthrough device designation for sustained monomorphic scar-related ventricular tachycardia...
Protaryx Medical has announced the completion of its first-in-human (FIH) study, assessing its advanced transseptal puncture technology.
Conducted on 3 December 2024 at Sanatorio Italiano de Asunción (Asunción, Paraguay), the study enrolled five patients under approval from the Research Ethics...
CathVision has unveiled the latest ECGenius system update—a release that brings essential enhancements to streamline workflows and boost procedural efficiency in electrophysiology (EP) labs, according to the company.
In a press release, CathVision notes that the electronic medical records (EMR)...
BTL Medical has announced the successful treatment of the first six patients in a first-in-human feasibility study using its next-generation catheter system designed to streamline atrial fibrillation (AF) ablation. This new technology is based on pulsed field ablation (PFA)—an...
Manipal Hospitals—one of the largest healthcare networks in India—has announced the successful insertion of an artificial intelligence (AI)-driven wireless injectable pacemaker.
The Aveir leadless pacemaker (Abbott), recently introduced in India, was implanted in a 65-year-old woman at Manipal Hospital...
New research suggests that cryoablation is a safe and effective approach to closing congenital patent foramen ovale (PFO) in patients with atrial fibrillation (AF) undergoing pulmonary vein isolation (PVI) as part of a single procedure. A study published in...
Electrophysiologists at the Texas Cardiac Arrhythmia Institute (TCAI) at St David's Medical Center (Austin, USA) recently became the first in the USA to use an all-in-one, high-density mapping and dual-energy ablation catheter (Affera ) for the treatment of persistent...
Late-breaking results from the PROMPT-AF, CRRF-PeAF and ARREST-AF trials were presented recently at the American Heart Association (AHA) Scientific Sessions (16–18 November 2024, Chicago, USA), shedding new light on the latest approaches to the management of patients with atrial fibrillation...
New research published in the European Heart Journal has shown that, among patients who have had a heart attack, it is “extremely difficult” to accurately predict those who will go on to suffer a sudden cardiac death—even in those with a...
Treatment with the Type 2 diabetes medication metformin, lifestyle changes, or a combination of both, did not improve atrial fibrillation (AF) burden or progression when compared with standard care, according to preliminary late-breaking science presented at the 2024 American...
In patients with atrial fibrillation (AF) or atrial flutter, COVID-19 vaccination appears to be “generally not associated with an increased risk of thromboembolic events”, as per the findings of a recent study led by researchers in South Korea. In...
Latest data from the Amulet investigational device exemption (IDE) trial, comparing the Amplatzer Amulet (Abbott) left atrial appendage occluder (LAAO) to the first-generation Watchman 2.5 (Boston Scientific) in patients with non-valvular atrial fibrillation (AF) at increased risk of stroke,...
Ceryx Medical has announced that the first patient has been enrolled in the RSA-Pace multicentre study at Waikato Hospital in Hamilton, New Zealand evaluating the safety of the Cysoni temporary pacing technology in the treatment of post-surgical heart failure...
Prescribing anticoagulation medications to adults younger than 65 years of age who have atrial fibrillation (AF) but no other risk factors for stroke does not reduce the risk of cognitive decline, stroke or transient ischaemic attack (TIA), according to late-breaking...
Three-year results from the OPTION clinical trial, comparing the Watchman FLX (Boston Scientific) left atrial appendage closure (LAAC) system to oral anticoagulation for stroke reduction in patients with non-valvular atrial fibrillation (AF), show that the device was associated with...
Ablation may be a better first-line treatment for heart attack survivors experiencing dangerous episodes of ventricular tachycardia (VT), according to late-breaking science presented recently at the American Heart Association (AHA) Scientific Sessions (16–18 November, Chicago, USA). This study has simultaneously...
A randomised trial comparing previously used and new pacemakers in patients with heart rhythm disorders has found reconditioned devices to be as safe and effective as their new counterparts, potentially offering affordable options for patients in low- and middle-income...
Two innovative electrocardiogram (ECG) products have gained new US Food and Drug Administration (FDA) clearances for monitoring cardiac arrhythmias—Fourth Frontier’s Frontier X Plus (FX+) and SmartCardia’s seven-lead (7L) patch. Both clearances were announced by the respective companies on 13...
In a paper published recently in the journal Chaos, researchers from Sergio Arboleda University (Bogotá, Colombia) and the Georgia Institute of Technology (Atlanta, USA) used an electrophysiological computer model of the heart’s electrical circuits to examine the effect of the...
An extra hour of physical activity every week may lower a person’s chance of developing the most common type of arrythmia by 11%, a new study shows.
Led by researchers at New York University (NYU) Langone Health (New York, USA),...
Stereotaxis has announced that physicians at the University of Kansas Health System (Kansas City, USA)—a system renowned for diagnosing and treating complex heart conditions—have successfully treated the first patients using the company’s advanced Genesis robotic magnetic navigation (RMN) system.
According...
A recent study published in JACC: Clinical Electrophysiology has placed emphasis on the importance of building research capacity for ambulatory rhythm monitoring in sub-Saharan Africa, where such services are currently scarce. Conducted in collaboration with the University of California...
Pulse Biosciences has announced the appointment of David Kenigsberg (Florida Heart Rhythm Specialists, Fort Lauderdale, USA) as chief medical officer (CMO) of Electrophysiology. In addition, Andrea Natale (St David’s Medical Center, Austin, USA) will become a medical advisor for...
Johnson & Johnson (J&J) MedTech has announced the US Food and Drug Administration (FDA) approval of the Varipulse pulsed field ablation (PFA) platform for the treatment of drug-refractory paroxysmal atrial fibrillation (AF).
“We have learned that, with PFA technology, mapping...
People with atrial fibrillation (AF) who have a stroke could benefit from receiving blood-thinning treatments—known as direct oral anticoagulants (DOACs)—at an earlier stage than is currently recommended, as per a new study led by researchers from University College London...
MedLumics recently announced that 10 patients have been treated in the company’s first-in-man clinical trial, which is ongoing and currently under protocol-dictated follow-up. The AblaView unipolar pulsed field ablation (PFA) system used in this trial features MedLumics’ unique, real-time,...
Boston Scientific Corporation announced today that it has entered into a definitive agreement to acquire Cortex, an Ajax Health company and privately held medical technology firm focused on the development of a diagnostic mapping solution that may identify triggers...
Adagio Medical has announced the completion of the first procedures in the FULCRUM-VT US Food and Drug Administration (FDA) pivotal investigational device exemption (IDE) study of the company’s ventricular tachycardia (VT) cryoablation system. The procedures were performed by William...
Abbott has announced a new, “first-of-its-kind” clinical trial designed to improve outcomes in patients with worsening heart failure who could benefit from advanced therapy options. The TEAM-HF trial—which is planned to enrol up to 850 patients across 75 sites...
Medtronic has announced US Food and Drug Administration (FDA) approval of the Affera mapping and ablation system with Sphere-9 catheter, an all-in-one, high-density (HD) mapping and pulsed field (PF) and radiofrequency (RF) ablation catheter for treatment of persistent atrial...
Boston Scientific has announced that the National Institute for Health and Care Excellence (NICE), has issued guidance recommending the use of the HeartLogic heart failure diagnostic as an option for managing heart failure in patients with cardiac implantable electronic...
The Heart Rhythm Society (HRS) board of trustees has unanimously approved the formation of Heart Rhythm Advocates—a new, shared-vision, cause-related non-profit 501(c)(4) advocacy organisation to complement the primary 501(c)(3) non-profit structure of the HRS.
According to the HRS, this announcement...
Boston Scientific Corporation has announced that it has received US Food and Drug Administration (FDA) approval of the navigation-enabled Farawave Nav ablation catheter for the treatment of paroxysmal atrial fibrillation (AF) and US FDA 510(k) clearance for its new...
Abbott announced today that it has achieved new, major milestones to support the company's growing suite of pulsed field ablation (PFA) solutions in electrophysiology.
These milestones include the early completion of enrolment in the VOLT-AF investigational device exemption (IDE) study...
Adjunctive coronary sinus isolation (CSI) as part of a de-novo ablation strategy does not confer any additional benefit compared to pulmonary vein isolation (PVI) plus roofline ablation in the treatment of high-burden atrial fibrillation (AF), as per a study...
A patient’s pattern of atrial fibrillation (AF) at their initial diagnosis does not influence their rate of AF recurrence, nor their AF burden, following a catheter ablation procedure—as per a multicentre study presented recently at the 2024 Asia Pacific...
Conformal Medical has announced the initiation of the GLACE Study, assessing the use of intracardiac echocardiography (ICE) imaging of the left atrial appendage (LAA) during appendage closure procedures with the next-generation CLAAS AcuFORM device in patients with non-valvular atrial...
The Health Policy and Regulatory Affairs Committee of the Heart Rhythm Society (HRS) has published a statement online in the Heart Rhythm Journal reaffirming the important role of electrophysiologists in cardiac ablation treatments for atrial fibrillation (AF).
Catheter ablation for AF is a...
Boston Scientific has announced receipt of Pharmaceuticals and Medical Device Agency (PMDA) approval in Japan for the Farapulse pulsed field ablation (PFA) system. The Farapulse PFA system—which is indicated for the isolation of pulmonary veins in the treatment of...
Medtronic has announced the presentation of clinical study results demonstrating a high rate of durable lesion formation with the PulseSelect pulsed field ablation (PFA) system in treating atrial fibrillation (AF). Invasive remapping conducted approximately two months post-ablation with the...
Boston Scientific recently announced late-breaking data from the MODULAR ATP clinical trial investigating the pacing performance of the company’s Empower leadless pacemaker. The data were delivered at the 2024 European Society of Cardiology (ESC) congress (30 August–2 September, London,...
Johnson & Johnson (J&J) recently shared that its medical technology businesses—including Cerenovus and Biosense Webster, as well as Abiomed, DePuy Synthes, and Ethicon—will now go by the name ‘Johnson & Johnson MedTech’.
This move “underscores J&J’s leadership in medical technology with...
Merit Medical Systems has announced that it recently signed a definitive asset purchase agreement to purchase Cook Medical’s lead management portfolio for a total cash consideration of approximately US$210 million.
Merit expects to fund the transaction through a combination of...
The treatment of atrial fibrillation (AF) via a novel, ‘single-shot’ spherical array approach to pulsed field ablation (PFA) has been found to achieve durable and safe creation of cardiac tissue lesions, translating into “good clinical efficacy”, at one-year follow-up....
Medtronic recently shared long-term results from the global EV ICD pivotal trial, reinforcing the performance and safety of the company’s extravascular implantable cardioverter defibrillator (EV-ICD) system, during a late-breaking clinical science presentation at the European Society of Cardiology (ESC)...
Quitting cigarettes can significantly lower a person’s risk of atrial fibrillation (AF) compared to those who continue to smoke, according to a study published recently in the Journal of the American College of Cardiology (JACC): Clinical Electrophysiology. The findings...
Boston Scientific has announced the appointment of Angelo Auricchio as chief medical officer (CMO) of the company’s Rhythm Management business in Europe, the Middle East and Africa (EMEA).
A renowned expert in cardiology and electrophysiology, Auricchio brings over 30 years...
Mass screening for atrial fibrillation (AF) using electrocardiography (ECG) together with a heart failure biomarker does not prevent ischaemic stroke or systemic in older adults—aged 75–76 years—over five years of follow-up. However, the biomarker in question may improve the...
Abbott has announced that, as of 6 September, it will be rolling out its Aveir dual-chamber (DR) leadless pacemaker system across the UK. In a press release, the company claims that Aveir DR is the “world's first” dual-chamber leadless...
In patients with persistent atrial fibrillation (AF), standard treatment with pulmonary vein isolation (PVI) ablation resulted in similar outcomes to more extensive ablation in other areas of the heart—as per results from the SUPPRESS-AF randomised controlled trial (RCT), which...
Adherence to atrial fibrillation (AF) guideline recommendations was found to be poor in clinical practice across Europe—but, a structured educational programme for healthcare professionals was able to improve implementation of heart rhythm control recommendations in patients. These are among...
B-Secur has announced the US Food and Drug Administration (FDA) clearance and launch of HeartKey Rhythm—a suite of electrocardiogram (ECG) algorithms and analytics designed to enhance clinical confidence, streamline industry efficiency, and improve outcomes for cardiac patients.
Powered by B-Secur’s...
Screening for atrial fibrillation (AF) using a wearable heart monitor for two weeks can identify older adults with this potentially dangerous abnormal heart rhythm, but does not prevent subsequent strokes, according to late-breaking research presented recently at the European...
As per late-breaking research presented recently at the 2024 European Society of Cardiology (ESC) congress (30 August–2 September, London, UK), cryoballoon (CB) ablation is as effective at reducing recurrences of atrial fibrillation (AF) and atrial tachycardia (AT) at one...
A catheter ablation procedure widely used to treat the most common heart rhythm disorder significantly reduces the burden of atrial fibrillation (AF), and results in clinically important improvements in symptoms and quality of life compared with a sham (placebo)...
Research presented at the 2024 European Society of Cardiology (ESC) congress (30 August–2 September, London, UK) will show that performing echocardiograms remotely using a 5G cellular network has similar accuracy to those performed in person by cardiologists.
“Comprehensive echocardiographic exam...
AtriCure has announced that the first patient has been treated with the AtriClip FLEX-Mini left atrial appendage (LAA) exclusion device, which recently received US Food and Drug Administration (FDA) 510(k) clearance.
The company describes the AtriClip FLEX-Mini as the smallest...
Biosense Webster has announced the enrolment completion of the Omny-IRE clinical trial.
Omny-IRE is a prospective, multicentre, non-randomised clinical trial that enrolled 188 patients in Europe and Canada to evaluate the safety and effectiveness of the Biosense Webster Omnypulse platform...
AtriCure has received an expanded indication for the AtriClip left atrial appendage (LAA) exclusion system in CE-marked countries in Europe.
The product is now indicated for use in patients at high risk of thromboembolism for whom left atrial appendage exclusion...
Work-related stress caused by job strain and an “imbalance between efforts applied versus rewards received” may increase the risk of developing atrial fibrillation (AF), as per new research published recently in the Journal of the American Heart Association.
Previous research has...
A study utilising nationwide healthcare data from across Denmark has indicated that, during catheter ablation procedures, conscious sedation (CS) may be linked to a higher risk of atrial fibrillation (AF) recurrence as compared to general anaesthesia (GA).
Writing in the...
The US Patient-Centered Outcomes Research Institute (PCORI) has announced the approval of funding awards totalling more than US$165 million for new patient-centred, comparative clinical effectiveness research (CER), with US$57 million being committed to two studies specifically focused on heart...
Pulse Biosciences has announced treatment of the first two patients in a first-in-human feasibility study using its novel nanosecond pulsed field ablation (nsPFA) cardiac surgery system for cardiac tissue ablation in the treatment of atrial fibrillation (AF).
Bart Van Putte...
BioCardia has submitted a 510(k) for approval of its patented Morph DNA steerable introducer sheath to the US Food and Drug Administration (FDA).
This product family is intended to provide a pathway through which medical instruments, such as balloon dilatation...
Adagio Medical has announced the completion of its business combination with ARYA Sciences Acquisition Corp IV—a special purpose acquisition company sponsored by an affiliate of Perceptive Advisors—to create a new firm focused on innovative technologies for the treatment of...
Element Science has announced the publication of results from its Jewel investigational device exemption (IDE) study—conducted at sites across the USA—in the Journal of the American College of Cardiology (JACC), as well as the recent publication of its Jewel EP...
The Hotwire radiofrequency (RF) guidewire device from Atraverse Medical has been used for the first time in clinical practice by world-renowned cardiac electrophysiologist Devi Nair at St Bernards Medical Center in Jonesboro, USA.
According to an Atraverse press release, the...
Ceryx Medical has announced the approval of a trial evaluating Cysoni-XT—a new temporary cardiac pacemaker aiming to boost recovery in heart failure.
The UK Medicines and Healthcare products Regulatory Agency (MHRA) recently approved Ceryx’s clinical trial authorisation application for the...
FibriCheck—a smartphone or smartwatch app capable of measuring a patient’s heart rhythm and detecting possible arrhythmias—has been cleared by the US Food and Drug Administration (FDA).
The clearance in the DHX category of smartphone electrocardiogram (ECG) devices means that US...
AtriCure has received regulatory approval from the National Medical Products Administration (NMPA) of China to market and sell several models of its AtriClip left atrial appendage (LAA) exclusion system.
The AtriClip device is designed to exclude and electrically isolate the...
The UK Medicines and Healthcare products Regulatory Agency (MHRA) has approved Ceryx Medical’s clinical trial authorisation application for the pilot respiratory sinus arrhythmia (RSA)-pace study of the Cysoni-XT system for post-cardiac surgery patients.
Cysoni-XT is a temporary cardiac pacemaker which...
Royal Brompton Hospital, part of Guy’s and St Thomas’ NHS Foundation Trust (London, UK) has become the first hospital in the UK to install the Magnetom Cima.X magenetic resonance imaging (MRI) system (Siemens Healthineers).
Magnetom Cima.X enables novel heart scanning...
Monitoring of heart rate and physical activity using consumer wearable devices was found to have clinical value for comparing the response to two treatments for atrial fibrillation and heart failure, the findings of a study published in Nature Medicine...
A patient in Cincinnati has become the first in the USA to receive a system that enables remote monitoring of his heart and congestion status, as well as self-adjustment of diuretics.
Vectorious Medical Technologies developed a miniature pressure sensor that...
This advertorial is sponsored by Boston Scientific
The arrival of pulsed-field ablation (PFA) has been heralded by many experts in the field of arrhythmia management as the most significant advance in the treatment of atrial fibrillation (AF) to have emerged...
This advertorial is sponsored by Biosense Webster
Biosense Webster founded the AFib Symposium in 2002. Now in its 22nd year, the annual event combines practical and scientific sessions on cutting-edge topics relevant to specialists in the treatment of atrial fibrillation...
Entries have opened for the second edition of the Global Cardiovascular Awards, celebrating innovation and excellence in the field of cardiovascular healthcare.
Returning on 13 March 2025, the second ever Global Cardiovascular Awards will feature 21 award categories recognising achievements...
Adona Medical has secured US$33.5 million in Series C financing, with the money to be used to further product development and initiate clinical use of the company’s adjustable interatrial shunt with integrated bi-atrial pressure monitoring.
The financing was led by...
Cordis recently announced that the US Food and Drug Administration (FDA) has granted premarket approval for its Mynx Control venous vascular closure device (VCD) for procedures with access sites from 6F-12F.
A press release details that the Mynx Control venous...
Pulsed field ablation (PFA) is safe for treating patients with common types of atrial fibrillation (AF), findings of the largest real-world registry of the technology to date have shown.
MANIFEST-17K is an international study that is the first to show...
Investment in a programme of clinical trials will underpin Boston Scientific’s strategy to expand the uptake of its Watchman left atrial appendage closure (LAAC) device, the company’s global vice president and general manager for the Watchman system, Angelo De...
Kardium, developer of the Globe mapping and ablation system for the treatment of atrial fibrillation (AF), has raised US$104 million in a new financing round.
The round is led by existing investor Fidelity Management & Research Company, together with follow-on...
Abbott has received CE mark in Europe for the Aveir dual chamber (DR) leadless pacemaker system.
The Aveir DR leadless pacemaker system utilises a new method of delivering dual chamber therapy as it is comprised of two unique devices—one that...
Results of the SPHERE Per-AF study, investigating the safety and efficacy of Medtronic’s Sphere-9 catheter, have been presented at the Heart Rhythm Society (HRS) annual meeting (16–19 May, Boston, USA), and have demonstrated the safety and efficacy of the...
Medtronic has announced that its investigational OmniaSecure defibrillation lead met its primary safety and effectiveness endpoints, exceeding prespecified performance goals in the global LEADR pivotal trial.
Results of the prospective, multicentre, single-arm, non-randomised, global clinical trial were presented at the...
Insights from the FARADISE registry, studying real-world outcomes from more than 1,000 patients to have undergone catheter ablation for paroxysmal and non-paroxysmal atrial fibrillation (AF) using the Farapulse pulsed field ablation (PFA) system (Boston Scientific) “reinforce the safety, efficacy...
Patients with previous paroxysmal atrial fibrillation (AF) who follow a Mediterranean diet enriched with extra-virgin olive oil after a catheter ablation procedure saw improved clinical outcomes when compared to those who freely selected their diet, research presented at the...
Six-month results from the ongoing pivotal MODULAR ATP clinical trial assessing the Boston Scientific’s mCRM modular cardiac rhythm management (CRM) system were presented at the Heart Rhythm Society (HRS) annual meeting (16–19 May, Boston, USA).
The system consists of the...
Late-breaking results from the pivotal phase of the admIRE pivotal clinical trial, alongside results from the VIRTUE study—both assessing the Varipulse (Biosense Webster) pulsed field ablation (PFA) system— were presented at the Heart Rhythm Society (HRS) annual meeting (16–19...
Atraverse Medical has announced US Food and FDA clearance of its Hotwire radiofrequency (RF) guidewire, a novel device that enables zero exchange left-heart access while also acting as a rail for catheter-based therapy systems.
This marks a significant step towards...
Adagio Medical has announced the completion of the first commercial procedures using its recently CE marked ventricular tachycardia (VT) cryoablation system and the US Food and Drug Administration (FDA) approval of the FULCRUM-VT pivotal IDE study of the VT...
Biosense Webster has announced the launch of the latest version of the its three-dimensional (3D) heart mapping system used in cardiac ablation procedures—Carto 3 system version 8.
The software features new modules including the Carto Elevate module and Cartosound Fam...
Corvia Medical has announced the publication of two-year echocardiographic data from its REDUCE LAP-HF II randomised clinical trial of the Corvia atrial shunt in JAMA Cardiology.
The study demonstrated favourable long-term effects of atrial shunting on heart structure and function...
Thermedical, a developer of advanced thermal-ablation systems to treat ventricular arrhythmias, announced today that it has completed a feasibility study utilising pulsed field ablation (PFA) therapy in combination with its SERF ablation system and Durablate catheter for treating ventricular...
Heart Rhythm Clinical and Research Solutions (HRCRS) and the Real World Evidence (RWE) Consortium have enrolled the first patient in the EVERCOOL AF study, a multicentre observational study evaluating the impact of proactive oesophageal cooling on patient outcome, throughput,...
Patients with heart failure who had an interatrial shunt inserted between the left and right atria did not see any significant benefits overall compared with those who received a placebo procedure after a median of 22 months follow-up, a...
Biotronik has announced the CE mark approval and first European implant of its latest insertable cardiac monitor (ICM)—Biomonitor IV.
The device features artificial intelligence (AI) for false positive reduction, and according to Biotronik is the only ICM on the market...
Three-month follow-up results from the SmartfIRE clinical trial, investigating Biosense Webster’s Thermocool Smarttouch SF dual energy catheter were presented in a late-breaking trial session at the European Heart Rhythm Association (EHRA) congress (7–9 April, Berlin, Germany).
Tom De Potter, an...
Clinical findings of the B3 study, assessing the impact of closed loop stimulation on the incidence of sub-clinical atrial fibrillation (AF) in sinus node disease patients, have been presented during a late-breaking trial session at the European Heart Rhythm...
Interim first-in-human clinical safety and efficacy results for Medtronic’s Sphere-360 investigational single-shot mapping and ablation catheter using pulsed field (PF) energy, for treatment of patients with paroxysmal atrial fibrillation (AF) were presented as a late-breaking clinical trial at the...
A randomised trial investigating preventive ablation of a potential arrhythmogenic substrate associated with coronary chronic total occlusion (CTO) in patients at high risk of ventricular arrhythmias (VAs) has found that this approach may reduce the risk of appropriate implantable...
A trial of technology using ultra-low temperature cryoablation (ULTC) has shown that the technique eliminated clinical ventricular tachycardia (VT) in 94% of patients.
The finding of the Cryocure-VT trial, which involved the use of Adagio Medical’s vCLAS catheter, were presented...
An international consensus statement on how to treat atrial fibrillation (AF) with catheter or surgical ablation has been published today in EP Europace and presented at the European Heart Rhythm Association (EHRA) 2024 congress (7–9 April, Berlin, Germany).
Lead author...
Boston Scientific has initiated the NAVIGATE-PF study of the Faraview software module when it is used to visualise and track the Farawave Nav pulsed field ablation (PFA) catheter for the treatment of patients with paroxysmal and persistent atrial fibrillation...
Automated external defibrillators (AEDs) were used in in 13 of nearly 1,800 cases of out-of-hospital cardiac arrest, even though many of the incidents occurred near a public AED, research to be presented at the American College of Cardiology’s (ACC’s)...
GE HealthCare has announced the launch of Caption AI artificial intelligence (AI)-driven software for Vscan Air SL, its dual-headed, handheld, wireless, point-of-care ultrasound imaging system.
With the addition of Caption AI, clinicians using Vscan Air SL handheld ultrasound will have...
Kardium has announced the successful completion of enrolment in the paroxysmal cohort of the pivotal PULSAR IDE (investigational device exemption) study of the Globe pulsed field ablation (PFA) system for the treatment of atrial fibrillation (AF).
This marks a milestone...
Viz.ai has announced a strategic collaboration with Medtronic to improve the coordination of cryptogenic stroke patient care between neurology and cardiology teams.
For stroke patients who are at risk of atrial fibrillation (AF) post-stroke and may need additional cardiac monitoring,...
High-power short-duration ablation results in significantly shorter procedure times without affecting procedural efficacy and safety among patients undergoing ablation for atrial fibrillation (AF).
This is according to the findings of a single-centre study investigating the impact of high-density mapping alongside...
Biosense Webster has submitted a premarket approval application (PMA) to the US Food & Drug Administration (FDA) for its Varipulse platform. The submission was supported by results from the admIRE study, a prospective, multicentre, non-randomised trial.
The Varipulse platform is...
Adagio Medical has announced CE mark approval of its ultra-low temperature cryoablation (ULTC) system for the treatment of monomorphic ventricular tachycardia.
The system consists of the upgraded cryoablation console, also capable of supporting atrial ablation procedures using commercially available iCLAS...
Abbott has announced it has received CE mark in Europe for its Assert-IQ insertable cardiac monitor (ICM).
Physicians can insert the Assert-IQ ICM under the skin of the chest to monitor a person's heart continuously and detect arrhythmias. After insertion,...
Following the recent CE approval of the Solia S lead for left bundle branch area pacing (LBBAP), Biotronik has announced the first complete conduction system pacing (CSP) system, now fully CE-approved for LBBAP.
The Biotronik CSP system comprises three components:...
People at risk should be tested for atrial fibrillation (AF) every time they attend a health appointment, according to results of the AFFECT-EU project. Patients at high risk of the disorder, such as those with heart failure or prior...
HeartBeam has announced that it has enrolled the first patients in the VALID-ECG pivotal study. The first patients were enrolled at Atlanta Heart Specialists (Atlanta, USA).
The VALID-ECG study will evaluate the performance of a 12-lead electrocardiogram (ECG) synthesised from...
The US Food and Drug Administration (FDA) has approved an additional indication for the weight loss drug semaglutide—marketed as Wegovy (Novo Nordisk) —to reduce the risk of major cardiovascular events such as death, heart attack, or stroke in adults...
Stereotaxis has announced that regulatory submissions were made recently in both Europe and the USA for the MAGiC catheter. These submissions follow initial clinical results in an ongoing trial.
Stereotaxis’ MAGiC catheter is a robotically navigated magnetic ablation catheter designed...
Haemonetics Corporation has entered into a definitive agreement to acquire Attune Medical, the manufacturer of the ensoETM proactive oesophageal cooling device, the only US Food and Drug Administration (FDA)-cleared temperature regulation device indicated for oesophageal protection during radiofrequency (RF)...
Six-month results from the PROACTIVE-HF pivotal trial, evaluating the Cordella pulmonary artery (PA) sensor (Endotronix) in New York Heart Association (NYHA) class III heart failure (HF) patients at risk of congestion, show that the trial met primary safety and...
BioCardia has announced interim results from the phase III randomised controlled trial of its CardiAMP autologous cell therapy in 110 randomised patients with advanced chronic heart failure at a mean 20-month follow-up, (CardiAMP HF).
Results showed reductions in heart death...
Volta Medical has announced CE mark for its Volta AF-Xplorer, an artificial intelligence (AI) companion that is cleared in the USA for the real-time manual or automatic annotation of spatiotemporal dispersions during AF and atrial tachycardia (AT) procedures.
Volta’s AF-Xplorer...
Biosense Webster has announced the receipt of European CE mark approval for the Varipulse platform for the treatment of symptomatic drug refractory recurrent paroxysmal atrial fibrillation (AF) using pulsed field ablation (PFA).
The Varipulse platform is comprised of the Varipulse...
FIRE1 today announced that it has completed patient enrolment in the US early feasibility study—FUTURE-HF2—of its FIRE1 system for remote heart failure monitoring.
The FIRE1 system is designed to directly measure a patient’s volume status by measuring the inferior vena...
A scientific statement published by the American Heart Association (AHA) in Circulation today acknowledges the potential “transformative” impact of the technology on cardiovascular medicine, but challenges remain in its development, the authors of the paper state.
The new scientific...
Researchers have detailed how artificial intelligence (AI) can be used to predict sudden cardiac arrest by studying patterns in electrocardiograms (ECGs) as well as differentiating between pulseless electrical activity and ventricular fibrillation.
The findings from researchers at Cedars-Sinai Health System...
VahatiCor has announced the treatment of the first patient with the A-Flux Reducer system, an interventional treatment for patients with angina or chest pain.
The implant is placed in the coronary sinus and is designed to provide more blood flow...
Biosense Webster has announced the commencement of patient cases with the investigational Laminar left atrial appendage elimination (LAAX) system as part of its pivotal investigational device exemption (IDE) study.
The first procedures, performed by Saibal Kar (Los Robles Health System,...
FineHeart has announced the successful filing of six new international patents for FlowMaker, a fully implantable device for the treatment of patients with advanced heart failure.
These patents increase the company’s international portfolio to 147 within 25 patent families, covering...
BiVACOR, a clinical-stage medical device company, has announced that US$13 million has been awarded from the Australian government’s Medical Research Future Fund (MRFF) grant through the Artificial Heart Frontiers Program (AHFP) to support BiVACOR’s total artificial heart programme and...
Reprieve Cardiovascular, a development stage company focused on pioneering an automated diuretic and fluid management approach for acute decompensated heart failure (ADHF) treatment, announced it has raised a total of US$42 million in series A financing.
The total round was...
X-trodes has announced that the US Food and Drug Administration (FDA) has granted 510(k) clearance for the company’s Smart Skin solution, marketed as X-trodes System M, a wireless wearable technology for advanced electrophysiological monitoring.
X-trodes’ Smart Skin is comprised of...
A person’s chance of surviving while receiving cardiopulmonary resuscitation (CPR) for cardiac arrest in hospital declines rapidly from 22% after one minute to less than 1% after 39 minutes, the findings of a US study published in The BMJ...
Pulse Biosciences has announced favourable findings from the 60-day post-procedure evaluations for four initial patients treated in the Company’s CellFX nanosecond pulsed field ablation (nsPFA) 360° cardiac catheter first-in-human feasibility study.
“The 60-day remap results for the initial patients treated...
This advertorial is sponsored by Biosense Webster
The field of catheter ablation for atrial fibrillation (AF) is evolving. The development of pulsed field ablation (PFA), as well as advances in established technologies such as radiofrequency (RF) ablation, help catheter ablation...
Biotronik has announced that it will solely supply the proprietary DX models for new single-chamber implantable cardioverter-defibrillators (ICDs) implants moving forward.
The move is being made in response to overwhelming recent clinical data demonstrating superior diagnostics and decreased complication risk...
Biosense Webster has announced support for two collaborative studies, VIRTUE and POLARIS aiming to better understand the use and workflows with the investigational Varipulse pulsed field ablation (PFA) platform for treating patients with diverse arrhythmias.
The VIRTUE study is evaluating...
Medtronic has announced new cost-effectiveness data from the STROKE AF clinical study, which showed that continuous monitoring with the Reveal LINQ insertable cardiac monitor (ICM) is significantly more cost-effective than the standard of care in ischaemic stroke patients with...
Twelve-month results from the inspIRE study, investigating predictors of success for pulmonary vein isolation with pulsed field ablation (PFA) using a variable loop catheter with 3D mapping integration have been presented as a late-breaking presentation at AF Symposium 2024...
Posterior wall ablation, a commonly used adjunct to pulmonary vein isolation (PVI), does not add any benefits for patients undergoing catheter ablation for persistent atrial fibrillation (AF), late-breaking research presented at AF Symposium 2024 (1–3 February, Boston, USA) has...
Findings of an investigation into whether ablation of non-pulmonary vein atrial fibrillation (AF) drivers identified by machine learning in addition to pulmonary vein isolation improves procedural outcomes have been presented at the AF Symposium 2024 (1–3 February, Boston, USA).
The...
Data from the first US comprehensive single-centre experience at St Bernard’s Medical Center and Arrhythmia Research Group (Jonesboro, USA), presented at a late-breaking session at the AF Symposium 2024 (1–3 February, Boston, USA) shows that left atrial appendage occlusion...
Boston Scientific has received US Food and Drug Administration (FDA) approval for the Farapulse pulsed field ablation (PFA) system.
The Farapulse PFA system is indicated for the isolation of pulmonary veins in the treatment of drug-refractory, recurrent, symptomatic, paroxysmal atrial...
Biotronik has announced the milestone achievement of 100,000 implanted single-chamber implantable cardioverter-defibrillators (ICDs) equipped with DX Technology.
Since the introduction of the technology in 2009, clinicians in more than 80 countries across all continents use DX ICDs.
“Reaching this significant milestone,...
Element Science has announced that it has received CE mark certification and UK Conformity Assessed (UKCA) marking for its novel patch-wearable cardioverter defibrillator (P-WCD) from its notified body, the BSI Group.
The Jewel P-WCD was designed to address limitations with...
The full shortlist for the inaugural Global Cardiovascular Awards—which will recognise the tireless work of individuals, teams and organisations to improve the life of sufferers of cardiovascular disease—has now been revealed.
An expert panel of judges has hand-picked finalists and...
Tributes have poured in from across the world of cardiology following the death of Bruce Wilkoff, a pioneer of cardiac electrophysiology and former president of the Heart Rhythm Society (HRS), who passed away early in January aged 69.
Wilkoff had...
Abbott has announced the first global procedures have been conducted using the company’s new Volt pulsed field ablation (PFA) system to treat patients with arrhythmias such as atrial fibrillation (AF).
Over 30 patients were recently treated in Australia as part...
Cardiac Dimensions, a developer of invasive treatment modalities to address patients suffering from heart failure with functional mitral regurgitation (FMR), has announced that interventional cardiologist Satya Shreenivas has joined the company as chief medical officer.
Shreenivas has led the structural...
Occlutech has announced that the US Food and Drug Administration (FDA) has approved the Occlutech ASD Occluder and Occlutech Pistol Pusher for the treatment of atrial septal defects (ASD).
With this approval, Occlutech will immediately begin commercialisation in an exclusive...
Orchestra BioMed Holdings has announced the first patient was randomised in the BACKBEAT pivotal study in late December 2023.
The BACKBEAT pivotal study will evaluate the efficacy and safety of atrioventricular interval modulation (AVIM) therapy (also known as BackBeat CNT),...
CardioFocus has announced the acquisition of Galvanize Therapeutics' Electrophysiology Technology division.
Notable assets included in the acquisition are the Centauri system pulsed electric field generator, which is CE marked and commercially available in the EU and the UK, and the...
People taking medical cannabis for chronic pain have a slightly increased risk of arrhythmia, according to research published in the European Heart Journal.
Recreational use of cannabis has been linked to cardiovascular disease but there has been very little research...
Biosense Webster has today announced approval from the Japanese Ministry of Health, Labour and Welfare (MHLW) for the Varipulse platform for the treatment of symptomatic drug refractory recurrent paroxysmal atrial fibrillation (AF) using pulsed field ablation (PFA).
The Varipulse platform...
GE HealthCare has entered into an agreement to acquire MIM Software, a provider of artificial intelligence (AI)-enabled image analysis and workflow tools across multiple care areas, including oncology, urology, neurology, and cardiology
GE HealthCare expects to leverage MIM Software’s imaging...
Stereotaxis has announced that the first patients have been successfully treated using its Magnetic Interventional Ablation Catheter, MAGiC.
Stereotaxis’ MAGiC catheter is a robotically navigated magnetic ablation catheter designed to perform minimally invasive cardiac ablation procedures. The first human procedures...
Medtronic has received CE mark for its Micra AV2 and Micra VR2, the next generation of its miniature, leadless pacemakers.
Micra AV2 and Micra VR2, the world’s smallest pacemakers, provide longer battery life and easier programming than prior Micra pacemakers,...
Endotronix has submitted a premarket approval (PMA) application for its Cordella pulmonary artery (PA) sensor system to the US Food and Drug Administration (FDA).
Cordella is a heart failure patient management platform that delivers proactive PA pressure data and non-invasive...
iRhythm Technologies has announced that its long-term ambulatory cardiac monitor, the Zio monitor ECG system, has received CE mark certification under the European Union’s Medical Device Regulation (MDR) from its notified body, the BSI Group.
The Zio monitor ECG system...
Pulse Biosciences has filed a premarket notification 510(k) to the US Food and Drug Administration (FDA) for its novel CellFX nanosecond pulsed field ablation (nsPFA) cardiac clamp.
Pursuant to Section 510(k), once the application has been accepted, the FDA will...
Allowing patients to eat before elective cardiac catheterisation posed no safety risk and benefited patient satisfaction and overall care, research published in the American Journal of Critical Care has shown.
Patients undergoing coronary artery catheterisation are typically required to take...
Boston Scientific has initiated the AVANT GUARD clinical trial to evaluate the safety and effectiveness of the Farapulse pulsed field ablation (PFA) system as a first-line treatment for persistent atrial fibrillation (AF), the only trial to study the use...
CardioRenal and CHU Grenoble Alpes have announced an innovation that allows chronically ill patients to measure their blood potassium levels autonomously and accurately.
The Tenor device enables patients to measure their blood potassium levels at home, similar to how diabetic...
LUMA Vision, developer of a novel four-dimensional (4D) cardiac imaging and navigation platform, has announced a total of US$22 million in new financing for the company.
The Series A3 financing added three new investors comprising an undisclosed multinational strategic investor,...
Biosense Webster has announced the first patient cases with the investigational dual energy Thermocool Smarttouch SF catheter took place as part of the SmartPulse pivotal study for treatment of paroxysmal atrial fibrillation (AF).
The first procedures were performed by David...
Which stories captured the attention of the electrophysiology community across 2023? Read our summary of the trending stories from across the Cardiac Rhythm News network throughout the year.
What were your highlights? Leave a reply in the comment box at...
Results of the National Cardiogenic Shock Initiative—a single-arm multicentre study assessing the feasibility and effictiveness of utilising early mechanical circulatory support using the Impella (Abiomed) device in patients presenting with acute myocardial infarction complicated by cardiogenic shock—have demonstrate a...
Individuals infected with COVID-19 are also at an increased risk of suffering from cardiac arrhythmias, research published online in European Heart Journal Open has concluded.
The authors of the study, Ioannis Katsoularis (University Hospital of Northern Sweden, Umeå, Sweden) and...
Smartwatches can help physicians detect and diagnose irregular heart rhythms in children, research published online in Communications Medicine has shown.
The finding comes from a survey of electronic medical records for paediatric cardiology patients receiving care at Stanford Medicine Children’s...
Medtronic has announced that the US Food and Drug Administration (FDA) has approved the PulseSelect pulsed field ablation (PFA) system for the treatment of both paroxysmal and persistent atrial fibrillation (AF).
This is the first PFA technology to receive FDA...
CathVision has announced the launch of ECGenius 3.1, an advanced version of its ECGenius system software.
The new version improves workflow and streamlines processes to accelerate adoption as electrophysiologists integrate artificial intelligence (AI) analyses into the electrophysiology (EP) lab, increasing...
Cardiovascular diseases, including ischaemic heart disease, stroke, heart failure, peripheral and aortic diseases, arrhythmias and valvular heart disease remain the leading global cause of death, and a major contributor to health loss worldwide, new data published in the Journal...
The cardiologist and researcher, Harlan M Krumholz (Yale School of Medicine, New Haven, USA), has been named editor-in-chief of the Journal of the American College of Cardiology (JACC), the flagship journal of the American College of Cardiology.
“The opportunities ahead...
Medtronic has announced the launch of its Penditure left atrial appendage (LAA) exclusion system in the USA.
The Penditure LAA Exclusion System is an implantable clip that comes pre-loaded on a single-use delivery system for use in left atrial appendage...
Procyrion has announced the enrolment of the first patients in the company’s investigational device exemption (IDE) pivotal trial.
The DRAIN-HF study will evaluate the Aortix percutaneous mechanical circulatory support (pMCS) technology in patients with acute decompensated heart failure (ADHF) who...
The American College of Cardiology (ACC) and the American Heart Association (AHA), with several other medical associations, have issued new guidelines for the prevention and management of atrial fibrillation (AF). The guideline was jointly published in the Journal of...
Johnson & Johnson MedTech has announced the completion of the acquisition of Laminar, a medical device company focused on eliminating the left atrial appendage (LAA) in patients with non-valvular atrial fibrillation (AF).
Johnson & Johnson MedTech acquired Laminar for...
Healthcare 21 (HC21) has gained national reimbursement for Catheter Precision’s VIVO 3D mapping system in the UK following it being granted inclusion in National Health Service (NHS) England's Specialised Services Devices Programme (SSDP).
NHS England’s SSDP, previously known as High-Cost...
Abiomed has announced that the first patient in the world has been enrolled in the landmark RECOVER IV randomised controlled trial (RCT).
The on-label, two-arm trial will randomise 548 patients to assess whether Impella support prior to percutaneous coronary intervention...
Ischaemic strokes in atrial fibrillation (AF) patients are less often disabling or fatal if they have been treated with left atrial appendage closure procedure (LAAC) compared to being treated with direct oral anticoagulants (DOACs), research published in JACC: Clinical...
The occurrence of atrial fibrillation (AF) after mitral valve surgery may be more harmful than previously thought, the authors of a research paper in the Journal of Thoracic and Cardiovascular Surgery (JCTVS) have stated.
Whitney Fu (University of Michigan Health...
A clinical trial to challenge the routine implantation of a defibrillator in myocardial infarction (MI) survivors with heart failure has enrolled its first patient. The PROFID EHRA trial is part of the EU-funded PROFID project, which aims to personalise...
Pulse Biosciences has announced the filing of a premarket notification 510(k) to the US Food and Drug Administration (FDA) for its novel CellFX nanosecond pulsed field ablation (nsPFA) percutaneous electrode.
The company’s percutaneous electrode is an image-guided needle designed to...
Medtronic has received CE mark for the PulseSelect pulsed field ablation (PFA) system and the Nitron CryoConsole.
The PulseSelect system is designed to treat atrial fibrillation (AF) effectively, efficiently, and safely, with a new ablation modality that uses pulsed electric...
Element Science has announced the successful completion and presentation of the results of the Jewel investigational device exemption (IDE) study, assessing the Jewel patch-wearable cardioverter defibrillator (P-WCD).
The study, which enrolled 305 patients, marks a significant milestone in the field...
Abbott has announced new late-breaking data that show advanced heart failure patients living with its HeartMate 3 left ventricular assist device (LVAD) who did not receive aspirin experienced fewer complications from bleeding and were associated with reduced hospital visits...
Analysis of outcomes of more than 17,000 patients receiving pulsed field ablation (PFA) using the Farapulse (Boston Scientific) device reinforce the safety profile of the system, investigators say, with no reported permanent phrenic nerve palsy, pulmonary vein stenosis or...
RCE Technologies, the developer of a non-invasive, instant measurement of cardiac proteins was selected as the winner of the annual Health Tech competition at the 2023 American Heart Association (AHA) Scientific Sessions (11–13 November, Philadelphia, USA).
The winning technology focuses...
Use of apixaban in patients with sub-clinical atrial fibrillation (AF) resulted in a lower risk of stroke or systemic embolism than aspirin, but a higher risk of major bleeding, results of the ARTESIA randomised trial, presented at the American...
Results of the SELECT clinical trial, presented at the American Heart Association’s 2023 Scientific Sessions (11–13 November, Philadelphia, USA), have shown that overweight or obese people without diabetes taking the drug semaglutide for more than three years had a...
Conformal Medical has announced positive one-year results from the company's CONFORMAL Early Feasibility Study (EFS).
William Gray (Lankenau Heart Institute, Wynnewood, USA) presented the Conformal CLAAS left atrial appendage occlusion (LAAO) device EFS one-Year TEE follow-up during a moderated abstract...
Patients with atrial fibrillation (AF) undergoing a transcatheter aortic valve implantation (TAVI) at the same time as a left atrial appendage occlusion (LAAO) procedure using the Watchman (Boston Scientific) device had similar outcomes when compared to patients getting TAVI...
Implicity has announced the results of a clinical study published in the Cardiovascular Digital Health Journal, reaffirming that its proprietary algorithm is highly effective at identifying patterns and classifying AF episodes into medically relevant events that require clinical action,...
Women with atrial fibrillation (AF) undergoing pulsed field ablation (PFA) have just as good outcomes as men according to a large-scale international study published in JAMA Cardiology.
This study is the first to compare sex outcomes for AF patients undergoing...
Biosense Webster has announced new findings from the Q-FFICIENCY study have been published in the Journal of Cardiovascular Electrophysiology, which the company says demonstrate improved control of atrial fibrillation (AF), relief of symptoms and overall quality of life, following...
Medtronic has received US Food and Drug Administration (FDA) approval for the Aurora EV-ICD MRI SureScan (extravascular implantable cardioverter-defibrillator) and Epsila EV MRI SureScan defibrillation lead to treat dangerously fast heart rhythms that can lead to sudden cardiac arrest (SCA).
The...
A research article in the journal Science has found that fibroblasts—scar-forming cells that reside in the scar tissue of an injured heart—directly play a role in promoting arrhythmia.
This finding holds promise for novel approaches to life threatening rhythm problems...
FEops has entered into a partnership with ConcertAI’s TeraRecon for the commercialisation of FEops HEARTguide’s for left atrial appendage (LAA) occlusion workflow, with a focus on the US market.
“We are thrilled to join forces with TeraRecon and integrate our...
Endotronix has received investigational device exemption (IDE) approval from the US Food and Drug Administration (FDA) for a subsequent multicentre study, PROACTIVE-HF 2, which will evaluate the company's Cordella sensor for pulmonary artery (PA) pressure-guided therapy.
The prospective, dual-arm trial...
This advertorial is sponsored by Boston Scientific
Physicians at Na Homolce Hospital (Prague, Czech Republic) have recently completed their 2,000th clinical case using the FARAPULSE (Boston Scientific) pulsed field ablation (PFA) system. The completion of the procedure marks an important...
Pulse Biosciences has announced a collaboration with CardioNXT, to support the company’s planned nanosecond pulsed field ablation (nsPFA) cardiac catheter first-in-human study focused on the treatment of atrial fibrillation (AF).
“We are excited to announce this collaboration and the progress...
Boston Scientific today launched the LUX-Dx II+ insertable cardiac monitor (ICM) system, describing it as a next-generation insertable monitor for long-term monitoring of arrhythmias associated with conditions such as atrial fibrillation (AF), cryptogenic stroke and syncope.
The system is designed...
Anumana has announced US Food and Drug Administration (FDA) 510(k) clearance for ECG-AI LEF, a breakthrough artificial intelligence (AI)-powered medical device to detect low ejection fraction (LEF) in patients at risk of heart failure.
“Anumana’s ECG-AI LEF fills an important...
This advertorial has been sponsored by GE HealthCare.
In this white paper Usman Siddiqui (Advent Health, Orlando, USA) discusses the reasons why the Prucka 3 with CardioLab Digital Recording System stands out among electrophysiology labs.
Featuring a robust ecosystem encompassing...
Research published in JACC: Clinical Electrophysiology has found a significant reduction in the rate of atrioesophageal fistuals (AEFs) during cardiac ablation of the left atrium when with the use of the ensoETM (Attune Medical) system when compared luminal oesophageal...
Medtronic has announced the results from two analyses demonstrating survival benefits and a reduction in life-threatening cardiac events with the use of implantable cardioverter defibrillators (ICDs).
The data provide real-world evidence supporting the use of Medtronic ICDs for indicated patients,...
Abbott has announced that the company's TactiFlex ablation catheter has been used for the first time in Canada at the Foothills Medical Centre in Calgary, to treat atrial fibrillation (AF).
The company describes the device as the world's first ablation...
Field Medical has closed its oversubscribed seed round with investments totalling US$14million.
The convertible note funding was led by private investors who were joined by multiple strategic investors. This financing will support preclinical-to-clinical development activities including first-in-human studies planned for...
Anthos Therapeutics has announced that the AZALEA-TIMI 71 Phase 2 study has been stopped early due to an “overwhelming” reduction in the composite of major and clinically relevant non-major bleeding in patients taking abelacimab compared to patients taking rivaroxaban.
The...
Biosense Webster has announced that the first cases have been performed using the investigational Omnypulse catheter as part of the Omny-IRE clinical trial. The first procedures were performed by Mattias Duytschaever at AZ Sint-Jan Hospital in Brugge, Belgium.
The Omny-IRE...
Younger atrial fibrillation (AF) patients are most likely to benefit from more personalised, magntetic resonance imaging (MRI)-guided ablation, according to a new analysis of the DECAAF II trial using artificial intelligence (AI). This was the conclusion of research presented...
Switching vitamin K antagonist (VKA) treatment to a non-vitamin K antagonist oral anticoagulant (NOAC) in frail elderly patients with atrial fibrillation (AF) is associated with more bleeding complications compared to continuing VKA treatment.
This is according to the findings of...
SmartCardia has received US Food and Drug Administration (FDA) clearance for its seven-lead real-time electrocardiogram (ECG) monitoring patch and cloud platform.
SmartCardia's 7L patch is cable-free, waterproof and can be used for continuous monitoring for up to 14 days.
“SmartCardia's 7L...
Adagio Medical has announced the first ultra-low temperature cryoablation (ULTC) procedure performed using the Adagio vCLAS catheter system in the USA as part of the FULCRUM-VT clinical trial. The procedure was performed at Banner - University Medical Center (Phoenix,...
Attune Medical has been granted de novo marketing authorisation from the US Food and Drug Administration (FDA) for its ensoETM device to reduce the likelihood of ablation-related oesophageal injury resulting from radiofrequency cardiac ablation procedures.
The FDA based its decision...
Stereotaxis has announced that physicians at Heart Centre Rigshospitalet of Copenhagen University Hospital successfully treated the first patients using Stereotaxis’ Genesis robotic magnetic navigation system. Rigshospitalet is among the first in Europe, and the only hospital in Denmark, to...
Vektor Medical has announced the release of a series of software enhancements to its AI-based non-invasive solution, vMap.
Designed to improve ablation outcomes and procedural efficiencies, the newly updated vMap software integrates additional automation and advanced visualisation features, the company...
Boston Scientific has received US Food and Drug Administration (FDA) approval for the latest-generation Watchman FLX Pro left atrial appendage closure (LAAC) device.
The device, which is indicated to reduce stroke risk in patients with non-valvular atrial fibrillation (NVAF) who...
Atrial fibrillation (AF) ablation is associated with lower rates of death, urgent heart transplantation or left ventricular assist device (LVAD) implantation compared with medical therapy in patients with end-stage heart failure, research presented in at the European Society of...
Biotronik has announced the first US implantation of the Amvia Edge pacemaker. The procedure was successfully completed by Raul Weiss at Mount Sinai Medical Center in Miami, Florida.
Amvia Edge is Biotronik’s next-generation pacemaker family, featuring MRI Guard 24/7, a...
The UK’s Medicines and Healthcare products Regulatory Agency (MHRA) has designated three new approved bodies to increase the country’s capacity to certify medical devices.
TÜV SÜD, Intertek, and TÜV Rheinland UK join the four current UK Approved Bodies, almost doubling...
Results of the first randomised trial comparing pulsed field ablation (PFA) to thermal ablation for the treatment of paroxysmal atrial fibrillation (AF)—ADVENT—have shown that PFA is as safe and effective at 12 months.
Findings of the study, which involves Boston...
GE Healthcare has announced the launch of two new tools for the cardiology market, coinciding with the European Society of Cardiology (ESC) congress (25–28 August, Amsterdam, The Netherlands).
The launches include Vscan Air SL, a dual-headed, handheld, wireless ultrasound imaging...
Oral anticoagulation in patients with atrial high rate episodes (AHRE) increases bleeding without reducing a composite outcome of stroke, systemic embolism or cardiovascular death.
This is the headline finding from the NOAH-AFNET 6 trial, presenting during a hot line trial...
Physical fitness is linked with a lower likelihood of developing atrial fibrillation (AF) and stroke, a study of more than 15,000 people, to be presented at the European Society of Cardiology (ESC) congress (24–28 August, Amsterdam, The Netherlands), has...
The British Heart Foundation (BHF) has appointed research leader and NHS consultant physician Bryan Williams as its first chief scientific and medical officer.
Williams will join the charity’s executive group in Autumn 2023 and succeed Sir Nilesh Samani (medical director)...
AstraZeneca’s Forxiga (dapagliflozin) has been approved in China for use in adults with symptomatic chronic heart failure.
The drug has previously been approved in China for heart failure patients with reduced ejection fraction (HFrEF) meaning that Forxiga is now approved...
Heart failure start-up Acorai has announced the initiation of a global clinical trial with the enrolment of its first patient in London, UK.
The Acorai heart monitor was designated as a breakthrough device by the US Food and Drug Administration...
An artificial intelligence (AI) model may be more efficient at detecting signatures of atrial septal defect (ASD) in electrocardiograms (ECG) than traditional methods.
This is according to investigators from Brigham and Women’s Hospital (Boston, USA) and Keio University (Tokyo, Japan),...
Research published in the journal Circulation: Cardiovascular Quality and Outcomes has highlighted the small number of clinical trials in cardiovascular medicine currently conducted in Africa.
The authors of the research letter, writing from Egypt and the USA, say that though...
CathVision has announced US Food and Drug Administration (FDA) clearance and commercial availability of the PVI Analyzer and Signal Complexity algorithms, part of the Cardialytics suite of artificial intelligence-powered analytics integrated into the ECGenius system.
According to a CathVision press...
Viz.ai has received a de novo approval by the US Food and Drug Administration (FDA) for its Viz HCM module, a hypertrophic cardiomyopathy (HCM) artificial intelligence (AI) detection algorithm.
The deployment of the algorithm is financially supported by a multi-year...
Ian Meredith is a well-known figure within the field of interventional cardiology, not only for a distinguished academic and clinical career but also his six-year spell as the executive vice president and global chief medical officer at Boston Scientific.
After...
Boston Scientific has received US Food and Drug Administration (FDA) approval for the POLARx cryoablation system.
The new system, which is indicated for the treatment of patients with paroxysmal atrial fibrillation (AF), features the POLARx FIT cryoablation balloon catheter, enabling...
Patient-tailored catheter ablation results in a significant reduction in atrial tachyarrhythmia (ATA) burden in shock-resistant persistent atrial fibrillation (AF) patients using implantable cardiac monitors (ICMs) implanted two months pre-procedure and conventional analysis may not capture the true impact of...
Ilika and Cirtec Medical have announced that they have signed a 10-year manufacturing licence to produce the Stereax range of mm-scale batteries at Cirtec’s facility in Lowell, USA.
Ilika, which is based in Romsey, UK, develops solid-state batteries, which can...
Biosense Webster has announced that several products in its cardiac ablation portfolio have received approval for a zero fluoroscopy workflow from the US Food and Drug Administration (FDA).
The products that can be used in this workflow include: Thermocool Smarttouch...
Researchers have presented a new method for assessing atrial remodelling in patients with atrial fibrillation (AF), based on the simultaneous assessment of electrical and contractile activity in the atria.
Details of the method have been set out in a paper...
The World Health Organization (WHO) has included a cardiovascular polypill—an all-in-one pill containing an antiplatelet, lipid lowering medication, and a blood pressure lowering and vascular stabilising drug (acetylsalicylic acid, ramipril, and atorvastatin)—in its List of Essential Medicines.
Developed by the...
Applying artificial intelligence (AI) to a single apical four chamber (A4C) view echocardiogram provides accurate information to detect heart failure with preserved ejection fraction (HFpEF), according to research published in JACC Advances.
The study, presented at the American Society of...
The combination of soaring heat and fine particulate pollution may double the risk of myocardial infarction (MI) mortality, according to a new study of more than 202,000 heart attack deaths in China. Findings of the study were published in...
This advertorial has been sponsored by GE Healthcare.
In this white paper Usman Siddiqui (University of Central Florida, Hunters Creek, USA) highlights reasons why the Prucka 3 with CardioLab digital recording system (GE Healthcare) stands out as the gold standard...
This post is sponsored by GE Healthcare
In this white paper, Usman Siddiqui (University of Central Florida, Hunters Creek, USA) highlight reasons why the Prucka 3 with CardioLab digital recording system (GE Healthcare) stands out as the gold standard in...
Biosense Webster has today announced the US launch of the Optrell mapping catheter with Trueref technology powered by the Carto 3 system.
The Optrell mapping catheter is a high-density diagnostic catheter, with small electrodes arranged in a fixed array formation...
Entries are now open for the inaugural edition of the Global Cardiovascular Awards, a recognition scheme highlighting the important contributions made by healthcare professionals and industry to improve outcomes for cardiovascular disease patients.
Held in association with Cardiovascular News, the...
Biosense Webster has announced that enrolment has been completed in the SmartfIRE study evaluating the safety and efficacy of its investigational Thermocool Smarttouch SF dual energy catheter and investigational Trupulse generator for the treatment of drug refractory symptomatic paroxysmal...
The use of embedded electrocardiogram (ECG) sensors in shopping trolley handles could effectively identify individuals with previously undiagnosed atrial fibrillation (AF). This is among the findings from the SHOPS-AF study, presented at the annual congress of the Association of...
The Heart Rhythm Society (HRS) has announced the appointment of Sami Viskin (Tel Aviv University, Tel Aviv, Israel) as the new editor-in-chief of Heart Rhythm from January 2024.
Heart Rhythm is the official journal of the HRS, the Cardiac Electrophysiology...
Medtronic is recalling all implantable cardioverter defibrillator (ICD) and cardiac resynchronisation (CRT-D) devices manufactured after 2017 with a glassed feedthrough, as there is a risk that the devices may deliver low or no energy output when high voltage therapy...
Stereotaxis has announced that the first patients have been successfully treated utilising Abbott’s EnSite X EP system with Stereotaxis’ robotic magnetic navigation system.
The combination of Abbott’s leading cardiac mapping system with Stereotaxis’ advanced robotic technology brings together highly detailed...
CathVision today announced its most recent financing round of US$9 million from investors.
The financing will help advance commercial operations driving adoption of the ECGenius system, the company's electrophysiology recording technology, and support the continued development of artificial intelligence (AI)-powered...
AccurKardia has announced that its flagship product, the AccurECG analysis system has been granted US Food and Drug Administration (FDA) 510(k) clearance.
AccurECG is a cloud-based, device-agnostic and fully automated electrocardiogram (ECG) interpretation software platform. The software includes beat-by-beat analysis,...
FIRE1 has announced that the first US patients have been successfully implanted with its FIRE1 system for remote heart failure monitoring in an early feasibility study (EFS).
The study will assess FIRE1’s novel solution to improve outcomes for heart failure...
Append Medical has raised US$4.35 million as part of an extended series A round, which will be used to support the company's first-in-human trials of Appligator.
Investors include participants from the first tranche of the series A round, as well as new investors,...
Sequana Medical NV has announced that the first patient has been enrolled in the MOJAVE study, a randomised controlled Phase 1/2a study in the USA, evaluating the safety and efficacy of the Company’s second-generation in diuretic-resistant DSR product (DSR...
Biotronik has announced US Food and Drug Administration (FDA) approval of its portfolio of Amvia Edge pacemakers and cardiac resynchronisation therapy pacemaker (CRT-P).
In a press release, Biotronik describes Amvia Edge as the market's smallest single-chamber MR conditional pacemaker and...
Researchers have developed a soft, fully bioresorbable, transparent microelectrode array (MEA) for monitoring heart disease and dysfunction.
Outlining the design, fabrication, characterisation and validation of the device in a paper published today in Science Advances, Zhiyuan Chen (The George Washington...
Abbott has announced that the US Food and Drug Administration (FDA) has approved the Aveir dual chamber (DR) leadless pacemaker system, described by the company as the world's first dual chamber leadless pacing system for the treatment of abnormal...
Patients who feel low when having a cardiac device implanted are more likely to stop taking their heart medications than those without depression, according to research presented at the annual congress of the Association of Cardiovascular Nursing and Allied...
Anumana has received breakthrough device designation from the US Food and Drug Administration (FDA) for its electrocardiogram (ECG) artificial intelligence (AI) algorithm designed to aid the early identification of cardiac amyloidosis.
The breakthrough device designation was granted to provide patients...
Philips, a global leader in health technology, has announced it has teamed up with Biotronik, a leading global medical device company with products and services that improve the lives of patients suffering from cardiovascular and endovascular diseases, to expand...
BioCardia, a developer of cellular and cell-derived therapeutics for the treatment of cardiovascular and pulmonary diseases, has announced that the US Food and Drug Administration (FDA) Center for Biologics Evaluation and Research (CBER) has completed review of the CardiAMP...
Findings of a retrospective cohort study assessing the long-term real-world safety and efficacy of leadless pacemakers implanted in patients throughout the Netherlands are “promising for longer-term data on leadless pacing”, the study’s authors suggest.
Writing in Heart Rhythm, Karel Breeman...
Procedure-related complications and mortality rates associated with catheter ablation of atrial fibrillation (AF) are low and have declined in the past decade, conclude the authors of a systematic review and pooled analysis, published in the Journal of the American...
CorWave has raised €61 million to fund its first industrial deployment and entry into clinical trials.
SPI fund, managed by Bpifrance on behalf of the French Government as part of France 2030, and leading family controlled entrepreneurial groups were the...
Nearly one-third of patients with an implanted device to prevent sudden death have anxiety in the first year while depression affects one in five, findings of a study in EP Europace.
“Implantable cardioverter defibrillators (ICDs) are effective at extending patients’...
The European Society of Cardiology (ESC) has announced the appointment of Jean-François Riffaud as its new chief executive officer. Riffaud will replace Isabel Bardinet, who will retire after 18 years at the ESC.
Riffaud is currently CEO of the French...
Arrhythmia Alliance has announced the launch of the first SVT Awareness Day (7 June), taking place during World Heart Rhythm Week (5‒11 June) to raise awareness of what it describes as a “little-known heart rhythm disorder that affects millions”.
Supraventricular...
The US Food and Drug Administration (FDA) has released draft guidance with updated recommendations for good clinical practices (GCPs) aimed at modernising the design and conduct of clinical trials.
In a statement, the regulator said that the updates are intended...
Researchers have developed an artificial intelligence (AI) model for electrocardiogram (ECG) analysis that allows for the interpretation of ECGs as language.
This approach can enhance the accuracy and effectiveness of ECG-related diagnoses, especially for cardiac conditions where limited data is...
Defibrillators are being used in just one in 10 cardiac arrests where they are available, according to research presented at the British Cardiovascular Society Conference (5–7 June, Manchester, UK).
The research drew upon data from the East of England Ambulance...
Conformal Medical has announced the successful completion of its series D funding round, raising a total of $35 million. The company is developing the CLAAS system, which it describes as a next-generation left atrial appendage occlusion (LAAO) technology
“SPRIG Equity...
Medtronic has announced that Ken Washington has been appointed chief technology and innovation officer.
Washington joins Medtronic from Amazon where he served as vice president and general manager of consumer robotics, and will lead technology development across industries including robotics,...
Kardium has announced today that the first US patients have been successfully treated in the PULSAR clinical study, using its Globe pulsed-field ablation system.
The PULSAR study will enrol over 400 patients for treatment at up to 35 sites in...
This article was sponsored by Biosense Webster
According to data published by Patrick M McCarthy (Feinberg School of Medicine, Chicago, USA) and colleagues in the Journal of Clinical Medicine, AF affects 1%—roughly 33 million—of patients in the western population, a...
New data presented from an investigator-sponsored European trial found managing indicated heart failure patients with Abbott’s CardioMEMS HF system resulted in a significant improvement in patient-reported quality-of-life scores as early as three months after use with the remote monitoring...
Medtronic has announced US Food and Drug Administration market clearance to apply the AccuRhythm AI algorithms to the Reveal LINQ insertable cardiac monitor (ICM), the predecessor to the LINQ II ICM, via cloud-based updates.
Medtronic anticipates the AccuRhythm AI algorithms...
An international clinical trial presented today at the European Stroke Organisation Conference (ESOC; 24–26 May, Munich, Germany) has demonstrated that—in people with ischaemic stroke and atrial fibrillation—anticoagulation can safely be started earlier than current guidelines recommend.
"Our study finally brings...
Preliminary results from the BIO-LIBRA study, assessing the outcomes of device-treated ventricular arrhythmias or mortality in patients with non-ischaemic cardiomyopathy (NICM) being treated with implantable cardioverter-defibrillators (ICDs) or cardiac resynchronisation therapy defibrillators (CRT-Ds) along with guideline-directed medical therapy (GDMT)...
Findings from a new clinical trial support use of implantable cardiac monitors (ICM) as a standard of care in managing patients with complex cardiac arrhythmias like atrial fibrillation (AF). The study will be presented today as a late-breaking clinical...
Results from a pivotal clinical trial found a leadless pacemaker can deliver cardiac resynchronisation therapy (CRT) among patients who were not able to be treated with conventional CRT and epicardial leads.
The novel WiSE CRT system removes the possibility of...
Abbott has announced that the US Food and Drug Administration (FDA) has approved the company's TactiFlex Ablation Catheter, Sensor Enabled, which features a flexible tip and contact force technology.
Used to perform an ablation procedure to treat atrial fibrillation (AF),...
A novel artificial intelligence (AI) model correctly identified patients at near-term risk of sustained ventricular tachycardia (VT) who could potentially benefit from preemptive interventions to prevent sudden cardiac death (SCD).
The AI-model utilises a single-lead electrocardiogram (ECG) screening tool that...
Results from a clinical trial, presented at the 2023 Heart Rhythm Society annual meeting (May 19–21, New Orleans, USA), indicate that overweight and obese patients with persistent and paroxysmal atrial fibrillation (AF) who lose weight prior to a catheter...
New data from three late-breaking clinical trials has demonstrated the safety and efficacy of pulsed field ablation (PFA) as a viable, non-thermal treatment option for atrial fibrillation (AF) and were presented at the 2023 Heart Rhythm (May 19–21, New...
Medtronic has announced findings from a secondary analysis of the PULSED AF study, demonstrating positive results for the Medtronic PulseSelect pulsed field ablation (PFA) system including atrial arrhythmia (AA) burden reduction, which correlated to improved quality of life and...
Stereotaxis has announced a global collaboration with Abbott to integrate Abbott’s EnSite X EP system with Stereotaxis’ Robotic Magnetic Navigation system.
The combination of Abbott’s cardiac mapping system with Stereotaxis’ advanced robotic technology brings together highly detailed real-time diagnostic information...
Late-breaking results from the AVEIR dual-chamber (DR) i2i Investigational Device Exemption (IDE) study, a large-scale study to assess Abbott’s Aveir dual-chamber leadless pacemaker, have been presented at the Heart Rhythm Society’s 2023 annual meeting (19–21 May, New Orleans, USA)...
Impulse Dynamics has today announced the completion of the first implantation for the INTEGRA-D clinical trial, designed to evaluate the safety and efficacy of two proven cardiac therapies combined—cardiac contractility modulation (CCM) and an implantable cardioverter defibrillator (ICD)—in a single...
MicroPort, a global developer in the field of cardiac rhythm management, announced today it has received US Food and Drug Administration (FDA) approval for its latest range of implantable pacemakers, Alizea and Celea, the longest-lasting pacemakers for their size on...
Biosense Webster—a stem of Johnson & Johnson MedTech—announced that data from the Q-FFICIENCY study was published in JACC: Clinical Electrophysiology. The study evaluated the safety and 12-month effectiveness of the QDot Micro catheter in paroxysmal atrial fibrillation (AF) ablation...
A recent study has found deep neural networks (DDNs)—a category of artificial intelligence (AI) algorithm—capable of forecasting cardiac pump function from standard angiography videos, providing accurate left ventricular ejection fraction (LVEF) predictions with strong correlations to echocardiographic LVEF measurements,...
AstraZeneca’s Farxiga (dapagliflozin) has been approved in the USA to reduce the risk of cardiovascular (CV) death, hospitalisation for heart failure (hHF) and urgent heart failure (HF) visits in adults with HF.
The approval by the Food and Drug Administration...
Biotronik announced today the latest addition to its cardiac rhythm management portfolio.
"We are excited to have received CE mark for our newest technology—the world’s first pacemakers and CRT-Ps approved for left bundle branch pacing. Amvia Sky and Amvia Edge...
Adagio Medical has announced the completion of enrolment in its Cryocure-VT trialof ultra-low temperature cryoablation for the treatment of monomorphic ventricular tachycardias.
Sixty patients with ischaemic and non-ischaemic cardiomyopathies underwent an endocardial ultra-low temperature cryoablation procedure using Adagio's vCLAS catheter...
Medtronic has received US Food and Drug Administration (FDA) approval of its Micra AV2 and Micra VR2 leadless pacemakers.
Micra AV2 and Micra VR2, the world's smallest pacemakers, provide longer battery life and easier programming than prior Micra pacemakers, while...
CathVision, a medical technology company developing innovative electrophysiology solutions to enhance clinical decision-making in the electrophysiology (EP) lab, today announced initial patient enrolment of a follow-on clinical study to demonstrate the value of the Signal Complexity algorithm led by...
Results from the MATRIX study show that the high detection accuracy of DX single-lead implantable cardioverter defibrillator (ICD) systems for atrial fibrillation (AF) episodes (99.7% for ≥1h episodes) in combination with the strong transmission performance of Biotronik home monitoring...
Effective management of depression through psychological therapy is associated with a lower likelihood of heart disease and stroke, according to research published today in European Heart Journal, a journal of the European Society of Cardiology (ESC).
“Our study suggests that...
Medtronic have announced the 12-month findings that supported the Affera Mapping and Ablation system CE Mark, demonstrating that the Sphere-9 catheter can successfully treat patients with either paroxysmal or persistent atrial fibrillation (AF) using a variety of ablation lesion...
Patients with severe COVID-19 requiring mechanical ventilation are 16 times more likely to develop ventricular tachycardia within six months compared to their peers without severe infection, according to research presented at the annual congress of the European Heart Rhythm...
A score based on four readily available clinical and imaging parameters identifies the heart failure patients who benefit most from atrial fibrillation (AF) ablation, according to late breaking science presented at the annual congress of the European Heart Rhythm Association...
An innovative three-step ablation approach including ethanol infusion of the vein of Marshall improves freedom from arrhythmias in patients with persistent atrial fibrillation (AF) compared to pulmonary vein isolation (PVI) alone, according to late breaking science presented at the...
A novel software tool set to improve the management of elderly atrial fibrillation (AF) patients with multiple conditions is being designed by the EU-funded and coordinated by the European Society for Cardiology (ESC) European Heart Rhythm Association (EHRA)-PATHS consortium....
High power electric vehicle chargers are safe for patients with pacemakers and defibrillators, according to a study published in EP Europace, a journal of the European Society of Cardiology (ESC) and presented at the annual congress of the European...
Spanish footballer, Iker Casillas, was awarded an European Heart Rhythm Association (EHRA) Gold Medal for services to cardiovascular health yesterday at the 2023 annual meeting.
In 2019, Iker, captain of the Spanish 2010 World Cup-winning team, suffered a heart attack...
Thirty-day electrocardiogram (ECG) monitoring in patients with hypertrophic cardiomyopathy (HCM) detects more arrhythmias than the standard 24 to 48 hours, according to late breaking science presented at the annual congress of the European Heart Rhythm Association (EHRA 2023, 16–18...
A region-wide study in more than 50,000 patients with atrial fibrillation (AF) has found reduced risks of stroke and transient ischaemic attack in those who started statins within a year of diagnosis compared with those who did not. The...
Stereotaxis has announced that over 1,000 cardiac arrhythmia patients have been treated using the Genesis Robotic Magnetic Navigation (RMN) system.
Genesis RMN, was first used in late 2020 following regulatory clearances in the USA and Europe. Since then, physicians at...
Boston Scientific has received CE mark for its POLARx FIT cryoballoon catheter for atrial fibrillation (AF) ablation.
According to the company, the device is the only cryoablation system that offers a dual diameter balloon size in one catheter to deliver an individualised...
Daytime napping for 30 minutes or longer is associated with an increased likelihood of developing atrial fibrillation (AF), according to research presented at European Society of Cardiology (ESC) Preventive Cardiology 2023, a scientific congress of the ESC.
“Our study indicates...
Wireless or leadless pacemakers, commonly implanted in adults, may be a safe and effective short-term option for children with slow heartbeats, according to new research published today in Circulation: Arrhythmia and Electrophysiology, a peer-reviewed journal of the American Heart...
SentiAR has closed a Series B financing round worth US$8.5 million. The financing was led by cultivate (MD) Ventures and joined by MedVenture Partners alongside several insider investors, including TechWald Holding, VCapital, QRM Capital, and Harmonix Fund. The funding...
Medtronic has announced the launch of MRI Care Pathway, a new system designed to streamline the process of completing magnetic resonance imaging (MRI) scans for patients with Medtronic MRI-compatible cardiac devices.
According to a company press release, research shows that...
Endotronix has announced its PROACTIVE-HF pivotal study has successfully completed enrolment.
The study is designed to evaluate the safety and efficacy of the Cordella pulmonary artery (PA) sensor, and the resulting data will support the premarket approval (PMA) application for...
Wearable devices such as smart watches could be used to detect a higher risk of developing heart failure and irregular heart rhythms in later life, suggests a new study led by University College London (UCL) researchers.
The peer-reviewed study,...
The Society for Cardiovascular Angiography & Interventions (SCAI) and the Heart Rhythm Society (HRS) have released an updated expert consensus statement on transcatheter left atrial appendage closure (LAAC).
In a press release, SCAI and HRS say that they prioritised the...
Results from the PADN-5 study, investigating the efficacy and safety of pulmonary artery denervation in the treatment of combined postcapillary and precapillary and pulmonary hypertension (CpcPH) related to left heart failure, have been presented at the Technology and Heart...
More than 40% of women report anxiety four months after a cardiac arrest compared with 23% of men, according to research presented at the scientific congress of the European Society of Cardiology (ESC) Acute CardioVascular Care 2023 (24–26 March,...
Patients with atrial fibrillation (AF) have been said to benefit from early rhythm control therapy, which reduces cardiovascular deaths, strokes, and other adverse outcomes by 20% compared to usual care, the European Society of Cardiology (ESC) has stated following...
Procyrion, a medical device company which aims to improve outcomes for patients with cardiac and renal impairment, announced today that use of its Aortix percutaneous mechanical circulatory support (pMCS) device led to rapid decongestion in a pilot study of...
Abbott has today announced new data that found monitoring patients remotely with haemodynamic pressure sensing technology, such as with its CardioMEMS HF System, can significantly improve survival in heart failure patients with reduced ejection fraction (HFrEF). The analysis is...
Vektor Medical has announced that use of the vMap arrhythmia mapping system during complex atrial fibrillation (AF) ablation was associated with significantly improved freedom from atrial arrhythmias compared with standard-of-care pulmonary vein isolation. Results will be presented at the...
Stereotaxis has announced the opening of a new robotic electrophysiology programme at Broward Health Medical Center in Fort Lauderdale, USA. Broward Health is among the first in the nation, and the first in Florida, to offer the latest Genesis...
A total of £10 million has been awarded to the Medicines and Healthcare products Regulatory Agency (MHRA)—an executive agency of the UK Department of Health and Social Care (DHSC)—to help bring innovative new medicines and medical technologies to UK...
HeartBeam, a cardiac technology company that has developed the three-dimensional (3D)-vector electrocardiogram (VECG) platform for heart attack detection today announced the strategic acquisition of all assets from LIVMOR, a digital health solutions company providing a patient-engaging remote monitoring system...
Kardium, a private medical device company that has developed the Globe pulsed field (PF) system for the treatment of atrial fibrillation (AF) using pulsed field ablation (PFA), announced today that the first patients have been successfully treated in its...
Acesion, the biotech developing first-in-class novel therapies for atrial fibrillation (AF), has today announced positive data from its phase two trial of AP30663, a first-in-class SK ion channel inhibitor for conversion of AF to normal sinus rhythm.
The trial enrolled...
Medtronic has announced today that it has received CE Mark for the Affera mapping and ablation system, which includes the Sphere-9 catheter and the Affera Prism-1 mapping software.
The full system creates a new paradigm in electrophysiology through the...
A recent analysis of a large nationwide database of patients with atrial fibrillation (AF) who underwent ablation has reported an independent association between being underweight and an increased risk of cardiac tamponade during ablation.
Cardiac tamponade is a potentially...
A retrospective cohort study investigating the rate of maternal characteristics associated with, and survival following, cardiac arrest during delivery hospitalisation, has found that older, non-Hispanic Black and low-income pregnant patients are disproportionately affected, but maintain better survival rates.
Published...
Using data from a large and “nationally representative” sample of the US population, a recent study is the first—the authors posit—to demonstrate racial differences in in-hospital outcomes after atrial fibrillation (AF) ablation in patients with heart failure (HF).
Published...
A recent study has provided new mechanistic insights identifying distinct atrial electrophysiological remodelling and fibrosis-associated conduction abnormalities favouring atrial fibrillation (AF) susceptibility in heart failure with preserved ejection fraction (HFpEF). The results from this proof-of-concept study support a correlation...
Biosense Webster has announced the first cases with its investigational Thermocool Smarttouch SF dual energy catheter have taken place as part of the SmartfIRE Study.
The first procedure was performed by Tom De Potter at OLV Hospital in Aalst, Belgium....
The European Union’s Council of Ministers has today adopted a resolution to extend the deadline for the certification of medical devices under the Medical Devices Regulation (MDR).
Producers of medical devices will have until 31 December 2027 for higher risk...
Results of the Pulsed AF Pivotal trial, investigating pulsed field ablation (PFA) treatment in patients with paroxysmal and persistent atrial fibrillation (AF), have been shared at the American College of Cardiology (ACC) 2023 Scientific Sessions (4 – 6 March,...
UC San Francisco (UCSF) is conducting a six-month clinical trial on hypertrophic cardiomyopathy (HCM) using Vivalink's Biometrics Data Platform. The study, consisting of 70 patients, will evaluate if regimented moderate intensity exercise improves overall exercise capacity and cardiac blood...
Early rhythm control one year after atrial fibrillation (AF) diagnosis has been found to be beneficial in preventing recurrent stroke in patients with incident AF and a prior history of stroke. Published in JACC: Clinical Electrophysiology, researchers suggest that...
Massachusetts Institute of Technology engineers have created a soft robotics-enabled three-dimensional (3D)-printed anatomical hydrodynamic system that can recreate the hemodynamic system of aortic stenosis (AS) and congenital defects in specific patient cases.
The researchers first took medical images of...
Biotronik has received CE mark approval for the Selectra 3D implant tools to include left bundle branch area pacing (LBBAP) in addition to His-bundle pacing (HBP).
Commonly referred to as conduction system pacing (CSP) these two approaches have emerged as...
Disabilities were underreported in clinical trial data and commonly used as a basis for exclusion from trial participation in an analysis of 80 recent trials involving cardiovascular outcomes, according to a study being presented at the American College of...
Atrial fibrillation (AF) ablation carried out in an inpatient setting is associated with higher rates of early mortality and subsequent complications when compared with outpatient AF ablation, a new study finds.
Referring to the Medicare fee-for-service (FFS) database, Mary-Jo...
CVRx has announced the preliminary topline results of the BeAT-HF—Baroreflex activation therapy for heart failure—postmarket randomised clinical trial.
The BeAT-HF postmarket phase of the multicentre, prospective, randomised controlled trial assessed 323 patients suffering from heart failure with reduced ejection fraction....
A new “red flag-raising” study has provided benchmark data on the safety of smart scales, watches and rings with bioimpedance technology for patients with cardiovascular implantable electronic devices (CIEDs). Discussing the research, principal investigator Benjamin Sanchez Terrones (University of...
Brainomix has announced its involvement in a new study sponsored by the University of Liverpool (Liverpool, UK) focused on post-stroke atrial fibrillation (AF). Sites with existing clinical deployments of Brainomix's e-Stroke platform will utilise the artificial intelligence (AI) system...
Medtronic has received CE mark for the Aurora extravascular implantable cardioverter-defibrillator (EV-ICD) magnetic resonance imaging (MRI) SureScan device and Epsila EV MRI SureScan defibrillation lead to treat dangerously fast heart rhythms that can lead to sudden cardiac arrest (SCA)....
A retrospective analysis of randomised clinical trials has found that one in five patients aged 70 years or older with cardiovascular risk factors are diagnosed with bradyarrhythmias when long-term monitoring for atrial fibrillation (AF) using implantable loop recorder (ILR)...
Kareem Abdul-Jabbar is working with Bristol Myers Squibb and Pfizer to raise awareness of atrial fibrillation (AF) and its symptoms as part of the No Time to Wait campaign.
In support of the campaign, Abdul-Jabbar will share his experience with...
University of Texas (UT) Southwestern Medical Center researchers have mapped gene control elements in specialised cardiac cells responsible for heartbeat regulation. The findings of the genome exploration study, published in the Journal of Clinical Investigation, provide insight into how...
Acutus Medical has announced results from the AcQForce Flutter study, evaluating use of the AcQBlate Force sensing ablation catheter and system in the treatment of right atrial typical flutter. Results were presented during a late-breaking clinical trials and first...
GE HealthCare has signed an agreement to acquire Caption Health, a developer of artificial intelligence (AI)-driven ultrasound technology.
The Caption Health technology—Caption AI—has been developed to detect signs of diseases such as heart failure in at-risk patients, potentially preventing hospitalisations...
In a study presented yesterday at the International Stroke Conference (ISC; 8–10 February, Dallas, USA), irregular heart rhythms were detected in roughly one in five people who survived an ischaemic stroke due to atherosclerosis and were continuously monitored for...
Biosense Webster has announced an update and late-breaking data from trials across radiofrequency ablation (RFA) and pulsed field ablation (PFA) at the 2023 AF Symposium (2–4 February, Boston, USA).
An analysis of the inspIRE-sponsored clinical trial evaluating the safety...
Abbott has today announced receipt of the CE mark for the TactiFlex ablation catheter, sensor enabled (SE), alongside US Food and Drug Administration (FDA) approval for an expanded indication of its FlexAbility ablation catheter, sensor enabled (SE).
In a press...
Among patients with heart failure with preserved ejection fraction (HFpEF) and a pacemaker, treatment with a moderately accelerated, personalised pacing rate resulted in an improved quality of life, N-terminal pro–brain natriuretic peptide (NT-proBNP) levels, and atrial fibrillation (AF) compared...
AtriCure has announced that the first patient was treated in the LeAAPS clinical trial, a prospective, randomised clinical trial to evaluate the safety and effectiveness of the AtriClip (Atricure) left atrial appendage (LAA) exclusion system for the prevention of...
Conformal Medical has announced the presentation of one-year data from the company’s initial left atrial appendage occlusion (LAAO) cases with its novel CLAAS system in patients under conscious sedation without general anaesthesia.
Vivek Reddy, (Mount Sinai Hospital, New York, USA)...
Researchers have found a correlation between periodontitis and fibrosis scarring to an appendage of the heart’s left atrium that can lead to atrial fibrillation (AF) in a sample of 76 patients with cardiac disease.
Periodontitis, a gum disease, can...
The prospective cohort study of 626 patients with non-magnetic resonance imaging (MRI)-conditional implantable cardioverter defibrillators (ICDs) suggests said ICDs remain suitable to treat detected tachyarrhythmias post-1.5-Tesla MRI.
Previous reports of adverse events have typically been noted during or immediately after...
BioVentrix, a privately held medical device company focused on the development of less invasive therapies to directly treat the dilated left ventricle and reverse the left ventricular remodelling process of progressive heart failure, today announced the US Food and...
tenacio, a company focused on digitising cardiovascular care to improve patient outcomes and reduce healthcare costs, has announced its OptiCor patient management platform is now commercially available following a completed review by the US Food and Drug Administration (FDA).
At...
Varian, associated with Siemens Healthineers, announced today that the US Food and Drug Administration (FDA) approved an investigational device exemption (IDE) for the pivotal RADIATE-VT clinical trial. The trial will be the first international, multi-centre, randomised controlled trial to...
An international study conducted by Leonard Yeo (National University Hospital, Singapore) and colleagues has found that the presence of atrial fibrillation does not modify the treatment effect of bridging intravenous thrombolysis (IVT) in ischaemic stroke patients undergoing a thrombectomy...
Ra Medical Systems announces the completion of its previously-announced stock-for-stock merger transaction with Catheter Precision, a company focused on the cardiac electrophysiology market.
MedTech veteran and Catheter Precision CEO and founder David Jenkins has joined Ra Medical’s board of directors...
Biobeat, a global leader in wearable remote patient monitoring solutions for the healthcare continuum, announced today that its wearable remote patient monitoring devices have received 510(k) clearance from the US Food and Drug Administration (FDA) to monitor stroke volume...
CardiacSense, a digital health company that developed the world's most advanced, medically certified wearable device for monitoring vital signs, announced receipt of US Food and Drug Administration (FDA) clearance of its CSF-3 watch for measuring beat-by-beat heart rate, and...
In their updated meta-analysis of 4,175 patients, Stergios Intzes together with a team from the Democritus University of Thrace, Alexandroupoli, Greece and the Heart Center, University of Leipzig, Germany, found P-wave duration (PWD) to be an independent predictor of...
HeartBeam, a cardiac technology company that has developed a 3D-vector electrocardiogram (ECG) platform for heart attack detection, has announced that a patent that enables generation of a synthesised 12-lead ECG by the HeartBeam AIMIGo credit card-sized device was issued...
A preclinical study demonstrating key features of the foam-based left atrial appendage closure device (CLAAS, Conformal Medical) has been published in the Journal of the Society for Cardiovascular Angiography and Interventions (JSCAI).
“This study validates the design features of Conformal's...
Cardiac Insight, a specialist in prescription-based wearable cardiac sensors and automated electrocardiogram (ECG) analysis software for cardiac arrhythmia diagnosis, has announced that researchers at Stanford Medicine selected the company’s Cardea SOLO wearable ECG system for a medical study entitled:...
Volta Medical has announced €36 million in new series B funding, bringing the total capital raised to over €70 million.
The company is developing artificial intelligence (AI) solutions to assist electrophysiologists in treating complex cardiac arrhythmias such as atrial fibrillation...
Anthos Therapeutics has announced that it has enrolled the first patient in LILAC-TIMI 76, a phase 3 study to evaluate the efficacy and safety of abelacimab in high-risk patients with atrial fibrillation (AF) deemed unsuitable for current anticoagulants by...
Johnson & Johnson has today announced it has completed its acquisition of Abiomed. Abiomed is now part of Johnson & Johnson and will operate as a standalone business within Johnson & Johnson’s MedTech segment.
“We are excited to officially welcome...
Primary care visits rise sharply in the weeks immediately preceding a sudden cardiac arrest, according to results from the ESCAPE-NET project. The project is backed by the European Heart Rhythm Association (EHRA) of the European Society of Cardiology (ESC)...
A cohort study of more than 4,500 persons without a history of atrial fibrillation (AF) or stroke has found that measuring left atrial mechanical function can improve stroke prediction. The findings are published in Annals of Internal Medicine.
AF is a...
The current model for randomised clinical trials must be redesigned for the modern age, according to the European Society of Cardiology (ESC), American Heart Association (AHA), World Heart Federation (WHF) and American College of Cardiology (ACC).
The call comes in...
Field Medical and CardioNXT have announced a strategic collaboration to provide the first-of-its-kind focal pulsed field ablation (PFA) therapy integrated with 3D mapping & navigation.
"PFA has generated much excitement in the treatment of atrial fibrillation due to an improved...
Biotronik announce the first patient enrolment in BIO-CONDUCT, an US Food and Drug Administration (FDA) approved Investigational Device Exemption (IDE) trial examining the use of the Biotronik Solia S pacing lead when implanted in the left bundle branch (LBB)...
With numerical simulations, researchers have demonstrated a new way to time weak electrical pulses that can stop certain life-threatening arrhythmias. Publishing their work in Chaos, by AIP Publishing, the group shows that timed pulses are successful in ending atrial...
Based in the USA, Weill Cornell Medicine has initiated a new clinical project together with the Mwanza International Trials Unit located in the Lake Zone of Tanzania with the aim of exploring the burden of cardiac arrythmias in adults...
Europe’s health commissioner, Stella Kyriakides, has announced that proposals to extend the transition period for the implementation of the European Union’s (EU) Medical Device Regulation (MDR) will be put forward in early 2023.
Kyriakides informed health ministers from the EU’s...
AstraZeneca has today announced that the Medicines and Healthcare products Regulatory Agency (MHRA) has granted a licence extension for dapagliflozin (Forxiga) in Great Britain for the treatment of symptomatic chronic heart failure (HF) in patients with HF and a...
The European Society of Cardiology (ESC) has urged European Union (EU) health ministers to act to prevent a “shortfall of essential medical devices for cardiovascular patients”, caused by challenges in the implementation of Europe’s Medical Device Regulation (MDR). Diagnostic...
Ultromics’ EchoGo Heart Failure, an artificial intelligence (AI) solution for echocardiography with the potential to revolutionise the diagnosis of heart failure with preserved ejection fraction (HFpEF), has received US Food and Drug Administration (FDA) clearance.
The system was developed by...
Medtronic has announced enrolment completion and final treatment in the SPHERE Per-AF, a US Food and Drug Administration (FDA) Investigational Device Exemption (IDE) trial. The study aims to evaluate the safety and efficacy of the Sphere-9 pulsed field (PF)...
A first-of-its-kind analysis on electrophysiological findings in patients with recurrent atrial tachyarrhythmia (ATa) following pulmonary vein isolation (PVI) using the novel pentaspline pulsed-field ablation (PFA) catheter reports a low incidence of pulmonary vein (PV) reconnection.
Shonta Tohoku (Cardiovascular Centre Bethanien,...
More scientific evaluation is needed to develop and validate consensus recommendations to ensure that benefits will outweigh risks for consumers who use smartwatches in screening themselves for possible atrial fibrillation (AF), according to the authors of an article published...
The Swiss Federal Assembly has voted in favour of accepting medical devices with US Food and Drug Administration (FDA) marketing authorisation in Switzerland.
A motion for ‘more freedom of action in the procurement of medical products for supply of the...
Abbott has been recognised by the Consumer Technology Association (CTA) with three Consumer Electronics Show (CES 2023; 5–8 January, Las Vegas, USA) Innovation Awards for multiple technologies that are said to be “advancing the health tech industry and improving...
An analysis of randomised controlled trials (RCTs) has found catheter ablation reduces mortality and heart failure hospitalisations in patients with atrial fibrillation (AF) and heart failure. Findings of the study were published in the journal Europace.
Carrying out a meta-analysis...
HeartBeam, a cardiac technology company that has developed the first and only 3D-vector electrocardiogram (VECG) platform for heart attack detection has announced that its patent for a 12-lead electrocardiogram (ECG) smartwatch-based monitor intended for detection of myocardial infarction (MI)...
Abbott has announced that Health Canada has approved the Aveir single-chamber (VR) leadless pacemaker.
The Aveir leadless pacemaker is implanted directly inside the heart's right ventricle via a minimally invasive procedure to treat slower-than-normal heart rates. Unlike traditional pacemakers, leadless...
A recent presentation by Paul J Wang (Stanford School of Medicine, Stanford, USA) at the American Heart Association (AHA) Scientific Session 2022 conference (5-7 November, Chicago, USA) outlined the results of the ENHANCE-AF study into a new shared decision-making...
Biosense Webster has announced the publication of data from a study comparing the risk of dementia in patients with atrial fibrillation (AF) who were treated with catheter ablation versus anti-arrhythmic drugs (AAD) in the American Heart Journal.
The study evaluated...
CVRx—the developer of the “world’s first” US Food and Drug Administration (FDA)-approved neuromodulation device to treat the symptoms of heart failure—has launched its new Barostim NEO2 implantable pulse generator (IPG).
The second-generation device reduces the size of the IPG by...
Ablation as a first-line treatment for atrial fibrillation (AF) disease is associated with significantly better clinical outcomes than starting with antiarrhythmic drugs.
These were the findings of the 36-month PROGRESSIVE-AF trial, presented by Jason Andrade (Vancouver General Hospital, Vancouver, Canada)...
WL Gore & Associates has announced that it is initiating the RELIEF clinical study—an investigational study to evaluate the safety and efficacy of transcatheter closure of patent foramen ovale (PFO) for the relief of migraine headaches utilising the Gore...
Screening to detect atrial fibrillation (AF) in older people would not only increase the chance of preventing stroke, it would also save money for the healthcare system and society. This is the conclusion from research published in the European...
iRhythm Technologies has announced a new health economic analysis of the mSToPS study, presented at the American Heart Association (AHA) scientific sessions 2022 (5–7 November, Chicago, USA).
The study evaluated the cost-effectiveness of screening for atrial fibrillation (AF) with Zio...
CathVision has announced the investigation of the Signal Complexity algorithm designed to visualise and quantify atrial fibrillation (AF) complexity parameters in patients with persistent AF. Ten patients have been treated to date at NYU Langone Health (New York, USA).
CathVision’s...
Acutus Medical has announced that it has achieved the first milestone under the asset purchase agreement of its left-heart access portfolio with Medtronic.
This triggers a US$20 million earnout payment from Medtronic to Acutus and allows Acutus to become an...
Boston Scientific has announced the European launch of the LUX-Dx insertable cardiac monitor (ICM) system, a long-term diagnostic device inserted under the skin of patients to detect arrhythmias associated with conditions such as atrial fibrillation (AF), cryptogenic stroke and...
Kardium has announced the successful first-in-human study of its next generation Globe pulsed field system to treat atrial fibrillation (AF) using pulsed field ablation (PFA) therapy.
Working with Vivek Reddy (Mount Sinai Hospital, New York, USA), and Petr Neužil...
Use of apixaban was associated with a lower risk of gastrointestinal bleed and similar rates of ischaemic stroke or systemic embolism, intracranial haemorrhage and all-cause mortality compared to other direct oral anticoagulants in patients with atrial fibrillation (AF), research...
Johnson & Johnson is to acquire Abiomed, it was announced today, after the two organisations entered into a definitive agreement under which Johnson & Johnson will acquire all outstanding shares of Abiomed through a tender offer.
The transaction will include...
Adagio Medical has announced its first pulsed field cryoablation (PFCA) procedure performed using the Adagio CryoPulse catheter system as part of the PARALELL (Pulsed field ablation and pulsed field cryoabLation in perrsistent atriaL fibriLlation) clinical trial.
The procedure was performed...
Researchers are to investigate the safety and efficacy of performing left atrial appendage occlusion in patients at high risk for atrial fibrillation (AF) who are undergoing cardiac surgery to prevent future stroke.
Richard Whitlock (McMaster University, Ontario, Canada) outlined the...
AccurKardia, a software company that provides clinical-grade, device agnostic, automated electrocardiogram (ECG) analytics, has announced it will collaborate with Mawi, a medtech provider of medical grade wearables in the cardiac space, to integrate its proprietary ECG analytics into Mawi’s...
In a large, population-based study of patients with atrial fibrillation (AF) and valvular heart disease, those receiving apixaban had a lower risk for ischaemic stroke or systemic embolism and bleeding, and a lower rate of intracranial or gastrointestinal (GI)...
Medtronic has received US Food and Drug Administration (FDA) approval for expanded labelling for the SelectSecure Model 3830 cardiac lead for left bundle branch area pacing.
In 2018, the FDA approved the lead for His-Bundle pacing, another form of conduction...
Catheter ablation of atrial fibrillation (AF) has favourable effects on cerebral blood flow, particularly in non-paroxysmal AF, research published in JACC: Clinical Electrophysiology.
The results may partially explain the association between cognitive decline and AF, Yoshihide Takahashi (Tokyo Medical and...
Biosense Webster has announced the European launch of the Heliostar radiofrequency balloon ablation catheter. Heliostar is indicated for use in catheter-based cardiac electrophysiological mapping of the atria and, when used with a compatible multi-channel radiofrequency generator, for cardiac ablation.
The...
Vektor Medical has announced study results that demonstrate a reduction in total procedural duration, fluoroscopy use, and cost after implementation of vMap, its cardiac mapping technology that uses only 12-lead electrocardiogram (ECG) data.
The research was presented by Avinash Toomu...
A real-world study of smartwatch electrocardiography (ECG) tools for the detection of atrial fibrillation (AF) has found that the use of such devices is “challenging” in patients with abnormal ECGs, and could be aided by better algorithms and machine...
Researchers have used artificial intelligence (AI) to evaluate patients’ electrocardiograms (ECGs) in a targeted strategy to screen for atrial fibrillation (AF). In the digitally-enabled, decentralised study, AI identified new cases of AF that would not have come to clinical...
Puzzle Medical Devices was announced as the winner of the Shark Tank innovation competition at the 2022 Transcatheter Cardiovascular Therapeutics meeting (TCT, 16–19 September, Boston, USA).
The company is developing a novel circulatory support device that is implanted percutaneously in...
This year marked the 20th edition of the Atrial Fibrillation Symposium (AFIB2022; 24–25 May, Copenhagen, Denmark) which, over its lifetime, has evolved to become one of the leading events in the calendar for atrial fibrillation (AF) specialists. Mark O’Neill...
Medtronic has announced that the Linq II insertable cardiac monitor (ICM) system has received 510(k) clearance from the US Food and Drug Administration (FDA) for use in paediatric patients over the age of two who have heart rhythm abnormalities...
Long-term follow-up of patients with non-valvular atrial fibrillation (AF) receiving the Amplatzer Amulet left atrial appendage occlusion (LAAO) device have shown continued safety and efficacy through three-years, results from the Amulet IDE trial shared at the 2022 Transcatheter Cardiovascular...
Implantable cardioverter defibrillator (ICD) information on YouTube is of low and highly-variable quality, according to research presented at the American College of Cardiology (ACC) Quality Summit 2022 (14–16 September, Los Angeles, USA).
As more patients turn to the internet for...
A study in Circulation: Arrhythmia and Electrophysiology has identified new-onset atrial fibrillation (AF) in one in 20 patients hospitalised with COVID-19.
Using data from the American Heart Association’s (AHA’s) COVID-19 Cardiovascular Disease Registry, researchers examined nearly 28,000 patients without a...
A new report from the American Heart Association’s (AHA’s) National Cardiac Implantable Electronic Device (CIED) Infection Initiative aims to improve adherence to guidelines on device explant following an infection.
The report is the result of a summit of key opinion...
Ra Medical Systems has entered into a merger agreement with Catheter Precision.
Under the terms of the agreement, Catheter Precision will become a wholly owned subsidiary of Ra Medical in a stock-for-stock reverse merger transaction.
If completed, the merger will result...
Haemonetics Corporation has received CE mark certification for its Vascade vascular closure and Vascade MVP venous vascular closure systems.
The CE marking will allow Haemonetics to engage in the next steps of country-specific entrance of both products into the...
Results of an analysis of Aktiia’s cuffless blood pressure (BP) monitoring system are being presented at the American Heart Association (AHA) Hypertension Scientific Sessions 2022 (7–10 September, San Diego, USA).
The analysis has been co-authored by experts from Barts NIHR...
Biotronik has announced a partnership with AliveCor, the developer of personal electrocardiogram (ECG) technology and services.
The partnership will bring together Biotronik’s Biomonitor injectable cardiac monitor with AliveCor's AI-enabled, clinically validated, medical-grade KardiaMobile 6L and KardiaMobile Card ECG technology.
Through this...
The US Food and Drug Administration (FDA) has granted an investigational device exemption (IDE) to Attune Medical to evaluate the company’s ensoETM in the reduction of oesophageal thermal injury during cardiac radiofrequency (RF) ablation procedures.
The study—Improving oesophageal protection during...
Vektor Medical has announced the publication of a peer-reviewed manuscript of the clinical data from a study evaluating the accuracy of its vMap technology in Circulation: Arrhythmia and Electrophysiology.
The system, which was cleared by the US Food and Drug...
AtriAN Medical has announced that it has completed enrolment of a second study using its selective pulsed field ablation (PFA) technology for the treatment of atrial fibrillation (AF).
The Neural AF-2 study has enrolled cardiothoracic surgery patients with paroxysmal AF,...
Biosense Webster has announced the release of the Octaray mapping catheter, developed for the mapping of cardiac arrhythmias including atrial fibrillation (AF). The catheter comes with TRUEref technology— a novel mapping reference electrode—powered by the Carto 3 version 7...
Boston Scientific has received US Food and Drug Administration (FDA) approval to expand the instructions for use labelling for the current-generation Watchman FLX left atrial appendage closure (LAAC) device to include a 45-day dual anti-platelet therapy (DAPT) option as...
Data from the PROMET and RELEASE studies provide important new insights into the safety and efficacy of rotational transvenous lead extraction (TLE), Christoph Starck (Berlin, Germany) and Peter Paul Delnoy (Zwolle, The Netherlands) tell Cardiac Rhythm News at the 2022 European...
LifeTech has announced that a US Food and Drug Administration (FDA)-approved investigator-initiated pre-market clinical trial of its proprietary LAmbre Plus left atrial appendage (LAA) closure system has obtained medical insurance coverage in the USA.
This is expected to facilitate the...
The addition of posterior wall isolation (PWI) does not improve arrhythmia outcomes compared with pulmonary vein isolation (PVI) alone in patients undergoing first time ablation for persistent atrial fibrillation (AF).
This is the concluding finding from the randomised, multicentre CAPLA...
Volta Medical has announced that peer-reviewed results from the company’s Ev-AIFib proof of concept study with its VX1 decision support software has been published in the Journal of Cardiovascular Electrophysiology (JCE).
The artificial intelligence (AI) software is designed to assist...
Researchers have developed a risk score to predict mortality for patients with atrial fibrillation (AF) who have undergone a successful transcatheter aortic valve implantation (TAVI) and have been discharged home.
The risk score is a product of the ENVISAGE-TAVI AF...
Medtronic has announced that it has completed the acquisition of Affera. The acquisition will expand Medtronic’s cardiac ablation portfolio to include its first cardiac mapping and navigation platform.
Among Affera’s offerings is the Affera Prism-1 cardiac mapping and navigation platform,...
Galaxy Medical has announced that it received the CE mark for its Centauri pulsed electric field (PEF) system and launched its commercial programme.
The approval allows the company to market the system for the treatment of paroxysmal atrial fibrillation (AF)...
Medtronic has announced that its investigational EV ICD system—a defibrillator with the lead placed under the breastbone, outside of the heart and veins—achieved a defibrillation success rate of 98.7% and met its safety endpoints in a global clinical trial.
Findings...
Screening for atrial fibrillation (AF) using a smartphone app more than doubled the detection and treatment rate in older people compared to routine screening, researchers behind the eBRAVE-AF trial have reported at the European Society of Cardiology (ESC) annual...
Results of one of the first and only randomised, blinded trials to assess the use of an artificial intelligence (AI) algorithm as an assessment tool for cardiac function suggest that AI improves evaluation when compared to assessment by a...
New guidelines for the management of patients with ventricular arrhythmias and the prevention of sudden cardiac death have been published today in the European Heart Journal and call for more automated external defibrillators (AEDs) to be placed in public...
Merit Medical has announced the launch of its SafeGuard Focus Cool compression device.
It is part of a broader cardiac portfolio that offers products and accessories necessary for cardiac rhythm management, electrophysiology, and lead extraction procedures as well as interventional...
Thermedical has announced that the US Food & Drug Administration (FDA) has approved an open-label, single-arm interventional clinical trial to evaluate the safety and efficacy of the Thermedical SERF ablation system with the Durablate catheter in people with ventricular...
Cannabis prescribed for chronic pain is associated with an elevated risk of heart rhythm disorders, according to research presented at the European Society of Cardiology (ESC) Congress (ESC 2022, 26–29 August, Barcelona, Spain).
Study author Nina Nouhravesh of Gentofte University...
Patients with recently diagnosed atrial fibrillation (AF) with a high burden of comorbidities should be considered for early rhythm control to reduce cardiovascular outcomes, whereas those with fewer comorbidities may have less favourable outcomes with this strategy.
These are among...
The National Institute for Health and Care Excellence (NICE) has issued draft guidance recommending KardiaMobile 6L for measuring cardiac QT interval in people having antipsychotic medication.
People taking antipsychotic medication may need testing for heart problems before starting treatment and...
A new scientific statement from the American Heart Association (AHA) provides guidance about sleep-disordered breathing and its association with the development of cardiac arrhythmia.
Published in Circulation, the analysis was authored by a panel led by Reena Mehra and Mina...
CathVision has secured US$7.2 million in funding from existing investors to help advance commercial operations driving adoption of the ECGenius system, the company's EP recording technology.
Funding will also support the continued development of artificial intelligence-powered analytic modules to provide...
A first-in-human multicentre trial using in-catheter, heated saline-enhanced radio frequency (SERF) ablation for patients with ventricular tachycardia (VT) has reported that the technique showed complete acute and satisfactory mid-term control of difficult VTs, which had failed one to five...
OSYPKA AG, a medical device company based in Rheinfelden, Germany, has received the CE mark under the European Union’s Medical Device Regulation (MDR) for a class III cardiac pacing electrode.
“This is a new milestone that is also valuable for...
The Heart Rhythm Society (HRS) has announced the full line-up of speakers and sessions for its first annual HRX meeting (8–10 September, San Diego, USA).
The conference offers clinicians, engineers, product developers, investors, entrepreneurs, non-profit organisations, and patient advocacy groups...
Projected rates of cardiovascular risk factors and disease will increase significantly in the USA by 2060, a study published in the Journal of the American College of Cardiology (JACC) has claimed.
Substantial increases in cardiovascular trends may contribute to a...
A project to develop genetic therapies to effectively cure cardiomyopathy has been announced as the winner of the British Heart Foundation’s Big Beat Challenge, a £30 million innovation project targeting unmet needs in heart disease.
CureHeart—a project involving researchers from...
Adagio Medical has announced the publication of the results of its Cryocure-2 ultra-low temperature cryoablation (ULTC) study in patients with atrial fibrillation (AF) in the Journal of American College of Cardiology (JACC), Clinical Electrophysiology.
The study prospectively enrolled 79 patients...
InCarda Therapeutics has announced enrolment of the first patient in the company’s pivotal phase 3 RESTORE-1 clinical trial of InRhythm (orally inhaled flecainide) in patients with newly-diagnosed atrial fibrillation (AF) or recurrent paroxysmal atrial fibrillation (PAF).
At an end-of-phase 2...
Acutus Medical has announced approval of its AcQMap high resolution imaging and mapping system and the AcQMap 3D imaging and mapping catheter in Japan.
Acutus’ International Alliance partner Biotronik has commenced training and early market development activities in collaboration with...
iRhythm Technologies has received US Food and Drug Administration (FDA) 510(k) clearance for its ZEUS (Zio ECG Utilization Software) system for the Zio Watch.
Produced in partnership with Verily, the ZEUS system combines deep learned algorithms with a cardiac arrhythmia...
Acutus Medical has announced that David Roman has been appointed president and chief executive officer, and member of the Board of Directors, effective immediately. The company also announced preliminary second quarter revenue results.
Roman, who joined the company as chief...
Around 80% of patients diagnosed with atrial fibrillation (AF) in the UK population are eligible for early rhythm control, an analysis of the EAST-AFNET 4 trial, published in Heart, has found.
Rhythm control therapy is typically delayed unless patients have...
Boston Scientific is collaborating with Telefónica España to provide ‘advanced healthcare technology’, including 5G, through its recently launched Remote Clinical Support Centre—RhythmCARE—which is intended to provide remote support to a number of hospitals in Europe, Africa and the Middle...
A research project at the University of Leeds (Leeds, UK) aiming to find an easier way to establish the optimum heart rate for heart failure patients with pacemakers, has received a £200,000 grant from Heart Research UK.
According to researchers,...
Stereotaxis has announced the CE mark submission for its MAGiC catheter, a robotically navigated magnetic interventional ablation catheter for minimally invasive cardiac ablation procedures.
MAGiC is used in conjunction with Stereotaxis’ robotic systems and designed to provide catheter precision, stability...
Abbott has introduced its EnSite X EP a cardiac mapping platform with EnSite Omnipolar Technology (OT) in Canada.
The platform allows for the creation of a 3D model of the patient’s cardiac anatomy in real-time, improving how physicians identify and...
Biotronik’s LiveSupport feature was used to remotely support two Biomonitor implantable cardiac monitor (ICM) implants shortly after launch. Both procedures were performed with a Biotronik representative in a remote location.
Developed in part as a response to the COVID-19 pandemic,...
Catheter ablation of atrial fibrillation (AF) was economically attractive compared with drug therapy in the CABANA (Catheter ablation versus antiarrhythmic drug therapy for atrial fibrillation) trial, investigators have reported in Circulation.
CABANA was an investigator-initiated, open-label, multicentre, randomised trial in...
Cardiac testing company CardiNor AS has announced that it has signed an exclusive agreement with IBL-America for sales of the CardiNor Secretoneurin ELISA test in the USA.
By initially targeting research institutes, clinical research organisations and pharmaceutical companies, the company...
HeartBeam is expanding the available patient population for its emergency department software technology solution. In evaluating the electrocardiogram (ECG) database for the clinical validation of HeartBeam’s platform technology, a significant portion of consecutive patients fell into the category of...
Acutus Medical has announced the completion of the first of two closings for its previously disclosed sale of the company’s left-heart access portfolio to Medtronic.
Additionally, Acutus Medical has also announced entry into a new longer-term credit facility with Deerfield...
People who develop an arrhythmia after surgery have an increased risk of subsequently being admitted to hospital with heart failure, according to a study of over three million patients published in the European Heart Journal. The study showed that...
Acutus Medical has announced the commercial launch of an expanded suite of left-heart access products to now include the AcQCross Qx system for use with the TruSeal and FXD delivery system for the Watchman left atrial appendage closure (LAAC)...
In patients treated with pulsed field ablation (PFA) for symptomatic paroxysmal atrial fibrillation (AF), the incidence of asymptomatic thromboembolic cerebral events or lesions detected by magnetic resonance imaging (MRI) was as low as 3%. This is according to the...
A study, published in the European Heart Journal–Digital Health, shows the predictive potential of a deep-learning model in identifying patients at risk of atrial fibrillation (AF) following monitoring with a 24-hour ambulatory electrocardiogram (ECG), despite no documented prior AF,...
Recommendations on how to use gene testing to prevent sudden cardiac death in athletes and enable safe exercise have been published in the European Journal of Preventive Cardiology.
“Genetic testing for potentially lethal variants is more accessible than ever before...
A national, multicentre registry investigating the safety and efficacy of laser lead extraction has reported high success- and low procedure-related complication rates associated with the procedure. This is the headline finding of the GALLERY (German laser lead extraction gallery)...
Conformal Medical has announced the launch of the CONFORM pivotal trial with the enrolment of the first patients at two US sites.
The investigational device exemption (IDE) study will evaluate the safety and efficacy of the company's CLAAS system compared...
Dual antiplatelet therapy (DAPT) after left atrial appendage occlusion (LAAO) using the Watchman FLX (Boston Scientific) device was comparable in terms of rates of death, stroke, bleeding or device-related thrombus when compared to use of aspirin with an anticoagulant,...
Incidence of ventricular arrhythmias can be linked to days when air pollution is high, according to research presented at Heart Failure 2022 (21–24 May, Madrid, Spain). The study was conducted in patients with an implantable cardioverter defibrillator (ICD), enabling...
AtriCure has announced that the first patient has been treated in the HEAL-IST trial, evaluating the safety and effectiveness of AtriCure’s Isolator Synergy Clamp for the treatment of drug-refractory patients diagnosed with inappropriate sinus tachycardia (IST). The first patient...
Abbott has announced that the US Food and Drug Administration (FDA) has approved the Aveir single-chamber (VR) leadless pacemaker for the treatment of patients in the US with slow heart rhythms.
The Aveir leadless pacemaker is implanted directly inside the...
A study has revealed the “global collateral damage” caused by the disruption to cardiac services caused by the COVID-19 pandemic. Writing in the European Heart Journal, researchers warn that problems with heart health will “...continue to accrue unless mitigation...
The use of a subcutaneous-implantable defibrillator (S-ICD, Boston Scientific) reduced the rate of major, lead-related complications by as much as 92% when compared to a transvenous device (TV-ICD), according to the investigators of the ATLAS trial, who presented their...
Findings of the US multicentre cardioneural ablation (CNA) registry were presented during a late-breaking clinical trial session at the Heart Rhythm Society’s 2022 annual meeting (HRS 2022, 29 April–1 May, San Francisco, USA), showing that ablation can be a...
Attune Medical has announced that the first patients have been enrolled in the IMPACT II— Improving Oesophageal Protection During Catheter Ablation for Atrial Fibrillation—study, evaluating the use of the ensoETM in cardiac radiofrequency ablation procedures.
Data from this study will...
inHEART has received US Food and Drug Administration (FDA) 510(k) clearance for its inHEART Models software suite that enables 3D visualisation and analysis of anatomical structures for pre-procedural planning and intraprocedural use.
With this clearance, inHEART will expand its commercial...
Endotronix has announced data from the SIRONA 2 clinical trial evaluating safety and efficacy of its Cordella pulmonary artery pressure sensor system in New York Heart Association (NYHA) class III heart failure patients.
The prospective, multicentre trial met all primary...
Findings of the EAST-AFNET 4 trial, examining the benefit of early rhythm control therapy in patients with newly diagnosed atrial fibrillation (AF), should influence future guidelines for the treatment of AF patients.
This was the message of Paulus Kirchhof (Hamburg,...
Results of a study presented at the Heart Rhythm Society’s 2022 annual meeting (HRS 2022, 29 April–1 May, San Francisco, USA) show that the success of intended same day discharge (SDD) improves over time for patients undergoing atrial fibrillation...
Use of an artificial intelligence (AI)-based app using electrocardiogram (ECG) signals recorded with an Apple Watch is able to identify left-ventricular dysfunction, research presented at the Heart Rhythm Society’s 2022 annual meeting (HRS 2022, 29 April–1 May, San Francisco,...
CathVision has announced US Food and Drug Administration (FDA) 510(k) clearance of the ECGenius electrophysiology recording system. ECGenius acquires high-fidelity, low-noise, cardiac electrograms to help electrophysiologists improve the diagnosis and treatment of complex atrial arrhythmias, including atrial fibrillation (AF).
Conventional...
Ceryx Medical has raised £3.8m in seed funding which will be used to commercialise its technology and commence the first-in-human clinical study of the cardiac rhythm management device, Cysoni, later this year.
The new funding involves Icehouse Ventures, The Development...
The Heart Rhythm Society (HRS) has announced the findings of three clinical trials demonstrating positive outcomes of conduction system pacing (CSP) for patients in need of cardiac resynchronisation therapy (CRT). The studies were presented as late-breaking clinical science at...
Results from a new clinical trial found aggressive blood pressure (BP) control reduced the risk of left-ventricular conduction disease. This study is the first to provide causal evidence that cardiac conduction disease is preventable, according to researchers. Findings were...
Abbott has announced US availability of its Amplatzer steerable delivery sheath, which is used with the company's Amplatzer Amulet left atrial appendage (LAA) occluder to treat people with atrial fibrillation (AFib) who are at risk of ischaemic stroke.
For patients...
Medtronic has announced new data from the STROKE AF clinical trial, showing that large and small vessel ischaemic stroke patients who receive short-term or intermittent monitoring using holter monitors and 30-day external monitors may not be optimally managed for...
CathVision has completed patient enrolment in the CathVision ECGenius System clinical evaluation study at the University of Vermont Medical Center (Burlington, USA). The study is the first in the USA to evaluate the safety and technical performance of ECGenius...
Medtronic has announced that Laura Mauri has been appointed as the company’s chief scientific, medical and regulatory officer. This appointment adds to Mauri's prior responsibilities as chief clinical and regulatory officer, aligning and integrating the company's scientific, medical, clinical research...
The use of high-power, short duration ablation for atrial fibrillation (AF) has the potential to speed up procedural times and increase the number of ablations that can be performed in a day.
This is the message of Teresa Strisciuglio (Clinica...
The pandemic saw a boom in the use of remote technologies and telehealth for patient monitoring and management, including for patients with heart failure—and many of these technologies look to have a role in the future, even despite a...
Medtronic has announced two AccuRhythm AI algorithms will be applied to LINQ II insertable cardiac monitors (ICM) through cloud-based updates in Europe later this spring.
AccuRhythm AI applies artificial intelligence (AI) to heart rhythm event data collected by LINQ II,...
Fitbit has received clearance from the US Food and Drug Administration (FDA) for a new photoplethysmography (PPG) algorithm to identify atrial fibrillation (AF). The algorithm will power a new irregular heart rhythm notification feature on Fitbit.
In a press release,...
Cardiologs has announced the results of a clinical study showing that its deep learning artificial intelligence (AI) software reduces inconclusive results returned by the latest Apple Watch ECG companion app—Apple ECG 2.0—while maintaining performance (sensitivity and specificity).
“Wearable devices, such...
Among the array of remote monitoring and digital arrhythmia management tools on display at the 2022 European Heart Rhythm Association annual meeting (EHRA 2022, 3‒5 April, Copenhagen, Denmark), Boston Scientific showcased its Heart Connect remote support system, which has...
FEops has announced that it received authorisation from the US Food and Drug Administration (FDA) for FEops Heartguide pre-operative planning of left atrial appendage occlusion (LAAO) with Abbott’s Amplatzer Amulet and Boston Scientific’s Watchman FLX device.
FEops Heartguide is a...
iRhythm Technologies has announced the results of three clinical research studies presented at the American College of Cardiology’s annual scientific sessions (ACC 2022, 2–4 April, Washington DC, USA).
According to iRhythm, the evidence further validates the Zio service as a...
Abbott has announced that the US Food and Drug Administration (FDA) has approved the Aveir single-chamber (VR) leadless pacemaker for the treatment of patients in the USA with slow heart rhythms. This marks significant advancement for patient care and...
Screening for atrial fibrillation (AF) should be integrated into flu vaccination and cancer screening programmes, according to a survey of general practitioners (GPs) conducted by the AFFECT-EU project and presented at the 2022 European Heart Rhythm Association annual meeting...
A simple electrocardiogram (ECG) can pinpoint hospitalised COVID-19 patients at high risk of death who might need intensive management, according to the authors of a study presented at the 2022 European Heart Rhythm Association annual meeting (EHRA 2022, 3‒5...
Overweight patients with atrial fibrillation (AF) are more likely to experience a return of the heart rhythm disorder after a corrective procedure than those of normal weight, according to research presented at the 2022 European Heart Rhythm Association annual...
Results of a large-scale, real-world analysis of US Centers for Medicare & Medicaid Services (CMS) data on the rates of guideline adherence and associated mortality in patients with cardiac implantable electronic device (CIED) infection show that only around one...
Use of cardiac magnetic resonance (CMR) may relevant role of CMR in risk stratification for sudden cardiac death after myocardial infarction, to late-breaking research presented at the 2022 European Heart Rhythm Association annual meeting (EHRA 2022, 3‒5 April, Copenhagen,...
Recommendations on the prevention and management of interference caused by medical procedures in patients with implanted electronic cardiac devices has been published today in EP Europace, and presented at the 2022 European Heart Rhythm Association annual meeting (EHRA 2022,...
The “exponential growth” in the use of digital devices for monitoring arrhythmias has led to the development of a new international consensus document. The paper, published today in EP Europace, will also be presented at the 2022 European Heart...
East End Medical has announced the first commercial use of its SafeCross transseptal radiofrequency (RF) puncture and steerable balloon introducer system. The system is designed to provide a predictable and safe solution for performing electrophysiology and structural heart interventions...
The launch of the latest KODEX-EPD cardiac mapping and navigation system is among the features Royal Philips will showcase at the 2022 European Heart Rhythm Association annual meeting (EHRA 2022, 3‒5 April, Copenhagen, Denmark), the company has revealed in...
GE Healthcare and AliveCor have announced a partnership to deliver medical-grade six-lead electrocardiograms (ECGs) taken by patients on an AliveCor KardiaMobile 6L ECG device outside of the hospital setting directly into GE Healthcare’s MUSE Cardiac Management System for physicians...
Researchers have developed a clinical algorithm that distinguishes between treatable sudden cardiac arrest and untreatable forms of the condition.
The findings, Journal of the American College of Cardiology: Clinical Electrophysiology, have the potential to enhance prevention of sudden cardiac arrest,...
Black and Hispanic individuals experiencing a cardiac arrest either at home or in public are substantially less likely to receive cardiopulmonary resuscitation (CPR) from a bystander compared to white individuals, according to a study presented at the American College...
Researchers want to leverage the centuries-old art of cutting paper into designs to develop a sensor sheet that can stretch and breathe alongside the skin while collecting electrocardiographic data, according to a press review. In Applied Physics Reviews, the...
Stereotaxis has announced that Poland’s National Institute of Cardiology in Warsaw has established the first robotic electrophysiology programme in the country with Stereotaxis’ Genesis robotic magnetic navigation (RMN) system. The National Institute of Cardiology is the only hospital in...
Research published in the journal BMC Medicine investigates the impact of kidney function in patients on antithrombotic therapy.
The study, a post hoc sub-group analysis focusing on recurrent bleeding events in the AFIRE (Atrial Fibrillation and Ischemic Events with Rivaroxaban...
Pulmonary vein-left atrial (PV/LA) volume ratio is a significantly better predictor power for atrial fibrillation (AF) recurrence than LA dimension in patients with normal or mild LA enlargement, with an association between PV/LA volume and the PITX2 gene. This...
A consensus document outlining design principles and outcome definitions for device-based therapies for hypertension has been published in the journal Circulation.
The development of the document has been led by the Hypertension Academic Research Consortium (HARC), which aims to create...
Pulsed-field ablation (PFA) and/or radiofrequency ablation (RFA) using a lattice-tip catheter can both isolate pulmonary veins (PVs) and create linear lesions efficiently and safely, with a high degree of lesion durability, and promising freedom from arrythmia recurrence. This was...
DiA Imaging Analysis Ltd, announced today a multi-year agreement with Change Healthcare. Change Healthcare's proven cardiology solutions, along with DiA's AI-based algorithms, will benefit echocardiologists and other imaging specialists by improving the efficiency of routine tasks and the effectiveness...
A new strategy using telehealth to monitor blood pressure at home for several months immediately after a stroke had a positive impact on patient engagement and blood pressure control among people who live in historically under-resourced communities, according to preliminary research presented at...
Hospitals in England are trialling remote monitoring technology from iRhythm Technologies—Zio XT—which is designed to aid the detection of arrhythmias supported by artificial intelligence.
Typically patients experiencing symptoms of an arrhythmia would need to go to hospital for 24 hours...
Royal Philips has announced new research evaluating mobile cardiac outpatient telemetry (MCOT) as a first-line diagnostic ambulatory monitoring solution with post-cryptogenic stroke patients.
The study determined that a 30-day continuous monitoring program using the Philips BioTel Heart MCOT patch, followed...
As a result of the COVID-19 pandemic, the increasing uptake of paediatric electrophysiology (EP) telehealth services has demonstrated clear ability to deliver healthcare for a pathologically and geographically diverse group of patients, who mostly report satisfaction with the service....
Portable electronic devices containing magnets may inhibit pulse generators for implanted cardiac devices (ICDs) and pacemakers according to new research published in Circulation: Arrhythmia and Electrophysiology.
The authors of the research, Corentin Féry (Institute for Medical Engineering and Medical Informatics,...
A recent systematic review and meta-analysis, published in the Journal of Interventional Cardiac Electrophysiology, has deemed same-day discharge following catheter ablation of atrial fibrillation (AF) to be safe and feasible.
According to a recent review, it is common practice...
Results from a new analysis assessing real-world outcomes with the Watchman FLX (Boston Scientific) left atrial appendage closure (LAAC) demonstrated a low rate of major adverse events at seven days following implant of the device.
The SURPASS analysis, which included...
Women remain underrepresented in leading cardiovascular clinical trials, corresponding with underrepresentation and a lack of gender diversity among presenters at scientific meetings, according to a review published in the Journal of the American College of Cardiology (JACC).
A previous analysis...
Wearable technology and digital health specialist Activinsights has announced a partnership agreement with the cardiovascular research organisation Cardialysis to promote the use of remote monitoring wearables as a tool for researchers to better understand the daily lifestyles of cardiac...
A review paper published in JACC: Clinical Electrophysiology explores the potential mechanisms linking stress and atrial fibrillation (AF), and the possible uses of stress reduction in the management of the condition.
Stress has been linked with poor health outcomes, though...
Historically, catheter ablation has been reserved for atrial fibrillation (AF) patients who have failed to respond to, or tolerate antiarrhythmic drug therapy, electrophysiologist Dhiraj Gupta (Liverpool Heart and Chest Hospital, Liverpool, UK) tells Cardiac Rhythm News. However, he explains,...
Both cryoballoon ablation (CBA) and hot balloon ablation (HBA) are effective treatment options for atrial fibrillation (AF), however, patients treated with HBA are more likely to receive touch-up ablation, with a longer procedure time. This was the main concluding...
Siemens Healthineers has announced the launch of Artis icono biplane—an angiography system with detectors optimised in size for integrated use in the cath lab.
The system offers new features for diagnosing and treating cardiac arrhythmia, coronary heart disease, and structural...
Abbott has announced that the US Food and Drug Administration (FDA) has approved an expanded indication for the company’s CardioMEMS HF system to support the care of more people living with heart failure.
With the expanded indication, an additional 1.2...
Medtronic has announced that the Freezor and Freezor Xtra cardiac cryoablation catheters are approved by the US Food and Drug Administration (FDA) to treat the growing prevalence of paediatric atrioventricular nodal reentrant tachycardia (AVNRT).
AVNRT is the most common form...
Fewer than half of UK citizens are aware of the link between arrhythmia and stroke, while around one in five people with a personal or familial risk of heart disease were unaware of how to check their pulse to...
Cardiologist and researcher Robert Califf has been confirmed as the commissioner of the US Food and Drug Administration (FDA). Califf returns to the role that he held between September 2016 and January 2017, having been confirmed in a vote...
A new timely practice—known as the “1-2-3-4-day” rule—for the administration of direct oral anticoagulants (DOACs) in patients after acute ischaemic stroke or transient ischaemic attack (TIA) is deemed feasible to decrease the risk of recurrent stroke, systemic embolism, and...
Boston Scientific has announced the close of its acquisition of Baylis Medical Company, a company that offers advanced transseptal access solutions as well as guidewires, sheaths and dilators used to support catheter-based left-heart procedures.
“The close of this acquisition allows...
Vektor Medical has announced that the University of California, San Diego Health (UCSD, San Diego, USA) is the first hospital system to offer the vMap arrhythmia mapping system.
The recently US Food and Drug Administration (FDA)-approved technology identifies potential arrhythmia...
CoreMap has announced the closure of a US$3 million series A milestone financing, with a US$20 million series B financing, which includes a diverse group of strategic, institutional, venture, and individual partners.
Pierre Jaïs of the Electrophysiology and Heart Modeling...
A collaboration between Ceryx Medical Limited and the scientists at the Auckland Bioengineering Institute (ABI) and the Universities of Bath and Bristol, have developed technology that they claim, could improve the outlook for patients with serious heart conditions.
Ceryx Medical's...
atHeart Medical has announced that it has received US Food and Drug Administration (FDA) approval for the start of the second phase of its ASCENT ASD US investigational device exemption (IDE) pivotal trial, evaluating the safety and efficacy of...
The placement of an interarterial shunt device—the Corvia atrial shunt (Corvia Medical)—did not reduce the total rate of heart failure events in a population of patients with heart failure with preserved or mildly reduced ejection fraction (HFpEF or HFmrEF).
This...
Abbott has announced the first patient implants of a dual-chamber leadless pacemaker system as part of its AVEIR DR i2i pivotal clinical study. The implant of Abbott's investigational Aveir dual-chamber leadless pacemaker represents a technological milestone for leadless pacing...
BioCardia has announced that the US Food and Drug Administration (FDA) has granted Breakthrough Device Designation for the CardiAMP cell therapy system for the treatment of heart failure.
CardiAMP cell therapy uses autologous bone marrow cells delivered to the heart...
A same-day discharge strategy for left atrial appendage occlusion (LAAO) could become the standard approach for this technique, according to the authors of a study published in the Journal of Invasive Cardiology, who concluded that the strategy is safe,...
AliveCor has announced the launch of KardiaMobile Card—an electrocardiogram (ECG) device the size of a standard credit card.
“After disrupting traditional ECG monitoring with our game-changing Kardia platform, we have now achieved the unprecedented milestone of creating the first-ever credit-card-sized...
Use of temperature-controlled ablation for the treatment of atrial fibrillation (AF) is effective and highly efficient, according to Jose Osorio—the director of electrophysiology at Grandview Medical Center in Birmingham, Alabama, USA. His comments follow the completion of a US-based...
The US Food and Drug Administration (FDA) has issued two final guidances providing recommendations for including patient perspectives in medical device clinical studies.
As per an FDA press release, the finalised version of the first of these two guidance...
Royal Philips has announced the introduction of a full-service, at-home, 12-lead electrocardiogram (ECG) solution for use in decentralised clinical trials. The clinical-grade solution is the most advanced patient-centric ECG offering within the company’s cardiac monitoring portfolio, pairing data readings...
A recent study has demonstrated 100% pulmonary vein isolation (PVI) using only pulsed field ablation (PFA), resulting in no PFA-related serious adverse events. This was the concluding finding of the PULSED AF (Pulsed-field ablation to irreversibly electroporate tissue and...
Contact-force (CF)-guided ablation to treat typical atrial flutter does not reduce recurrent atrial arrhythmia at 12 months follow-up, in comparison to ablation blinded for contact force. This was the main conclusion of a randomised controlled trial (RCT), findings of...
Acute alcohol consumption is associated with a higher risk of atrial fibrillation (AF), a study published in Nature Cardiovascular Research has concluded
The study is the first to show an association between increased drinking and hospital visits for AF in...
Royal Philips is supporting the American Heart Association’s (AHA’S) effort to generate awareness among cross-disciplinary specialties and improve survival rates from cardiovascular implantable electronic device (CIED) infections.
With the support of Philips, the American Heart Association’s national CIED infection initiative...
According to a press release, AtriAN Medicals clinical data from the Neural atrial fibrillation (AF) study was presented by Vivek Reddy (Mount Sinai Hospital, New York, USA) at this year's renowned annual AF symposium 2022 (13—15 January, New York...
MicroPort CRM has recently received the pharmaceuticals and medical devices agency (PMDA) Japanese regulation agency approval for the Alizea range of portable pacemakers. The devices are equipped with bluetooth technology for streamlined remote monitoring when paired with MicroPort CRM's...
Abbott has received clearance from the US Food and Drug Administration (FDA) for the EnSite X EP system with EnSite Omnipolar Technology (OT), a new cardiac mapping platform available in the USA and across Europe that is designed to...
Medtronic has received approval from Japan's Ministry of Health, Labor and Welfare for the sale and reimbursement of the Micra AV Transcatheter Pacing System (TPS), and the company will launch the product this month.
This approval expands the number of...
Medtronic has announced that it has entered into an agreement to acquire medical technology company Affera.
Affera designs and manufactures cardiac mapping and navigation systems and catheter-based cardiac ablation technologies, including a differentiated, focal pulsed field ablation (PFA) solution, for...
Khaldoun Tarakji, associate section head of cardiac electrophysiology at the Heart, Vascular, and Thoracic Institute at Cleveland Clinic (Cleveland, USA) has joined Medtronic as vice president and chief medical officer (CMO) of the company's Cardiac Ablation Solutions (CAS) Operating...
The National Institute for Health and Care Excellence (NICE) has issued Medical Technologies Guidance (MTG) recommending KardiaMobile (AliveCor) as an option for detecting atrial fibrillation (AF) in patients with suspected paroxysmal AF, who present with symptoms such as palpitations...
Endotronix has announced that the US Food and Drug Administration (FDA) has granted approval for an amendment to the company's PROACTIVE-HF study, a pivotal Investigational Device Exemption (IDE) trial investigating the Cordella Pulmonary Artery (PA) Pressure Sensor, shifting the...
Adagio Medical has announced first-in-human cases of pulsed field cryoablation (PFCA) for the treatment of atrial fibrillation (AF).
The cases were performed at Medicover Hospital, Warsaw, Poland by Pawel Derejko, head of the Department of Cardiology at Medicover and Atul...
Volta Medical has announced the introduction of VX1 artificial intelligence (AI) software at three US hospitals. New York Presbyterian Queens, Northwell Health’s Lenox Hill Hospital both in New York, and Ascension St. Vincent’s Riverside, Jacksonville, have been named as...
Doctors at St David's Medical Center in Austin, USA recently implanted a new neurostimulator technology in patients to help treat advanced heart failure—and are among the first physicians in the USA to do so, according to a press release.
The...
Affera has announced that the first patient was treated in the recently approved SPHERE PerAF trial, a US Food and Drug Administration (FDA) Investigational Device Exemption (IDE) pivotal randomised trial, to evaluate the safety and effectiveness of the Affera...
FAST BioMedical has announced the publication of additional results from the EMPAKT CHF (Estimating versus Measuring Plasma Volume and Kidney Function in Acute Decompensated Congestive Heart Failure) clinical trial in the European Society of Cardiology (ESC) Heart Failure journal....
Kardium has announced the completion of successful first-in-human (FIH) procedures with the Globe pulsed field system.
Vivek Reddy of Mount Sinai Hospital (New York, USA) and Petr Neužil of Na Homolce Hospital (Prague, Czech Republic), performed the procedures in Prague,...
Implicity has announced US Food and Drug Administration (FDA) clearance for a novel medical algorithm that analyses ECG data from implantable loop recorders (ILRs).
“This is an important step for Implicity as we expand in the US market,” said Arnaud...
LifeTech Scientific Corporation has announced that it has extended its agreements with Medtronic to further the strategic collaboration on the HeartTone domestic pacemaker project and to start the collaboration on domestically-made MRI-conditional pacemakers. Together with MRI-conditional leads, these products...
Stereotaxis has announced the publication of the results of a prospective, multicentre study comparing the incidence of silent cerebral embolism (SCE) in atrial fibrillation (AF) patients undergoing robotic cardiac ablation or manual in Frontiers in Cardiovascular Medicine.
The study evaluated...
New research suggests that implantable cardioverter defibrillators (ICDs) are not used as frequently for females and patients of colour, despite the increased use of ICDs in the management of patients with hypertrophic cardiomyopathy (HCM), over recent decades.
In patients with HCM,...
Royal Philips has announced an expansion of its ultrasound portfolio with new imaging tools and features for cardiology, designed to “increase diagnostic confidence and workflow efficiency”. Philips will be showcasing these developments through its participation in the EuroEcho 2021...
SyMap Medical has announced the completion of a financing round which will be used to accelerate global clinical trials for the company’s renal mapping and selective renal denervation systems.
The financing round of nearly US$100 million was led by Primavera...
One in 10 junior doctors training to be cardiologists in the UK say they have been bullied, the results of a survey published online in the journal Heart indicate. Women and those who qualified in medicine outside the UK...
Restore Medical has announced preliminary results of the first-in-human clinical trial of ContraBand, a breakthrough treatment for patients suffering from congestive heart failure. The minimally-invasive, catheter-delivered implants are used to treat patients with chronic left ventricular failure.
ContraBand has been...
Boston Scientific has announced the start of the MODULAR ATP clinical trial to evaluate the safety, performance and effectiveness of the mCRM modular therapy system. The mCRM system consists of two cardiac rhythm management (CRM) devices intended to work...
Persistent opioid use following a cardiac implantable electronic device (CIED) procedure is common, according to a study published in Circulation, which reports that 12% of patients receiving a CIED are consistently using pain medication in the months after the...
Canon Medical has announced the introduction of the Precise IQ Engine (PIQE) deep-learning reconstruction (DLR) and SilverBeam filter.
PIQE, which Canon describes as super-resolution deep-learning reconstruction technology for cardiac computed tomography (CT) scans, delivers “exceptional” cardiac CT image quality by...
Acutus Medical has announced the initiation of AcQForce PFA-CE, a clinical study that will evaluate the safety and performance of the company’s focal force sensing pulsed field ablation (PFA) catheter and system in combination with its novel noncontact 3D...
Cardiologs announced that it has received 510(k) clearance from the US Food and Drug Administration (FDA) to use its AI-powered cardiac diagnostics platform for paediatric cardiology.
The expanded authorisation was granted based on an analysis of the company's improved deep...
The advent of pulsed field ablation (PFA) will revolutionise the treatment of atrial fibrillation (AF), Tom de Potter (Aalst, Belgium) tells Cardiac Rhythm News, discussing the future of the treatment of the condition.
PFA utilises a controlled electric field to...
European Society of Cardiology (ESC) guidance for the diagnosis and management of cardiovascular disease during the COVID-19 pandemic has been published in the European Heart Journal.
The guidance brought together a large number of key opinion leaders at the outset...
Cardialen has received approval from the US Food and Drug Administration (FDA) for an investigational device exemption (IDE) to begin a clinical trial of its Multipulse Therapy (MPT) to treat paroxysmal and persistent atrial fibrillation (AF).
MPT is delivered as...
Women diagnosed with breast cancer are significantly more likely to develop atrial fibrillation (AF) compared to women without cancer, according to research published in the European Heart Journal. These patients have a three-fold increased risk of death from either heart...
Results of the Fitbit Heart study, presented during a late-breaking science session at the American Heart Association’s Scientific Sessions 2021 (AHA 2021; 13–15 November; virtual), suggest that the consumer fitness tracking watch, Fitbit, was capable of detecting undiagnosed atrial...
HeartVista has received US Food and Drug Administration (FDA) 510(k) clearance to deliver its AI-assisted One Click magnetic resonance imaging (MRI) acquisition software on Siemens Healthineers MRI scanners.
The use of cardiac MRI, also known as cardiac magnetic resonance (CMR),...
Ahead of the American Heart Association’s 2021 annual scientific sessions (AHA 2021, 13–15 November, virtual), Cardiac Rhythm News speaks to the meeting’s programme committee chair Manesh Patel (Duke University School of Medicine, Durham, USA) about some of the highlights...
Data from the global Leadless II IDE study evaluating the Aveir (Abbott) leadless pacemaker in patients with certain abnormal heart rhythms show that the device met its prespecified primary safety and efficacy endpoints .
The findings were presented today in...
Adults with type 1 or type 2 diabetes and atrial fibrillation (AF) are less likely to notice irregular heartbeat symptoms, more likely to have a lower quality of life (QoL), and experience more co-existing health conditions than people with...
Caption Health and Ultromics have announced a strategic partnership to accelerate cardiovascular disease detection and treatment for more patients in more accessible care settings. Together, the companies will jointly offer the Caption AI software platform alongside Ultromics’ EchoGo deep...
Vektor Medical has announced US Food and Drug Administration (FDA) 510(k) clearance for its novel computational electrocardiogram (ECG) mapping system, vMap.
vMap is designed to map potential arrhythmia sources associated with stable or unstable arrhythmias anywhere in the heart—including all...
Patients who went into cardiac arrest in the cardiac cath lab were more likely to survive to hospital discharge than those who had a cardiac arrest in the intensive care unit (ICU), yet less likely to survive than those...
Medtronic has announced its ambition to achieve net zero carbon emissions by fiscal year 2045 across its operations and value chain to accelerate efforts to combat climate change. The announcement comes amidst the 2021 United Nations Climate Change Conference...
Royal Philips has signed an agreement to acquire Cardiologs, a developer of cardiac diagnostics using artificial intelligence (AI) and cloud technology. Cardiologs will further strengthen Philips’ cardiac monitoring and diagnostics offering with innovative software technology, electrocardiogram (ECG) analysis and...
Findings of the SWISS-APERO trial, presented at the Transcatheter Cardiovascular Therapeutics annual meeting (TCT 2021, 4–6 November, Orlando USA and virtual), comparing the Amulet (Abbott) and Watchman FLX (Boston Scientific) left atrial appendage closure (LAAC) devices, show similar rates...
Left atrial appendage occlusion (LAAC) remains non-inferior to non vitamin-K-antagonists (NOACs) for the prevention of major cardiovascular, neurological or bleeding events in patients with non-valvular atrial fibrillation (AF) at long-term follow-up.
These are four-year findings of the PRAGUE-17 clinical trial,...
South Korean artificial intelligence (AI) developer Vuno has announced that the Korean Ministry of Food and Drug Safety (KFDA) has designated the company's AI-based electrocardiogram (ECG) analysis software Vuno Med-DeepECG as a breakthrough device. Using deep learning, the software...
Researchers from Massachusetts General Hospital (MGH, Boston, USA) have reported the detection and characterisation of thrombosis using fibrin-targeted positron emission tomography and magnetic resonance in a first-in-human study published in JACC: Cardiovascular Imaging.
Atrial fibrillation (AF) can cause clots to...
Research presented at the American College of Rheumatology’s annual meeting (ACR Convergence, 3–9 November, virtual) shows that hydroxychloroquine does not appear to be associated with QTc interval prolongation, an irregularity of the electrical activity of the heart that places...
A pragmatic clinical trial—CHANGE AFib—will determine whether early treatment with the antiarrhythmic drug dronedarone improves cardiovascular and long-term outcomes in patients presenting with first-detected atrial fibrillation (AF). The trial represents a collaboration between the American Heart Association (AHA) and...
CathVision has announced the first patient enrolments in the PVISION multicentre clinical study, investigating the automated assessment of pulmonary vein isolation (PVI) using the company's artificial intelligence (AI) algorithm, a PVI analyser, in combination with the ECGenius system, CathVision's...
An artificial intelligence (AI)-based computer algorithm created by researchers at the Mount Sinai Health System (New York, USA) has been able to learn how to identify subtle changes in electrocardiograms (ECGs) to predict whether a patient was experiencing heart...
Ethnic and racial minority cardiologists report career satisfaction but face professional discrimination and exclusion. This was the main finding of the American College of Cardiology (ACC) professional life survey, published in the Journal of the American College of Cardiology...
Stereotaxis has announced publication of a study in the International Journal of Cardiology: Heart & Vascular demonstrating superior safety and efficacy of Stereotaxis’ robotic technology to treat atrioventricular reentry tachycardia (AVRT) and atrioventricular nodal reentry tachycardia (AVNRT) in paediatric...
Baylis Medical Company has reached an agreement to sell its cardiology business to Boston Scientific.
The acquisition, for an upfront payment of US$1.75 billion, subject to closing adjustments, will see Baylis Medical Company and its cardiology products transition to Boston...
Medtronic has announced the publication of a study demonstrating, through the use of a continuous rhythm monitoring device, that atrial fibrillation (AF) is directly and transiently associated with ischaemic stroke. The findings were published online in JAMA Cardiology.
This retrospective...
Haemonetics Corporation has announced that the Vascade MVP venous vascular closure system is has received a US Food and Drug (FDA) indication for same-day discharge following atrial fibrillation (AF) ablation.
The same-day discharge labelling was granted following the conclusion of...
The US Food and Drug Administration (FDA) has informed healthcare providers of recent information about the potential for differences in procedural outcomes between women and men undergoing implant of a left atrial appendage occlusion (LAAO) device.
The FDA is evaluating...
Abbott has announced that the US Food and Drug Administration (FDA) has approved the company's Amplatzer Talisman PFO Occlusion System to treat people with a patent foramen ovale (PFO)—a small opening between the upper chambers of the heart—who are...
Biosense Webster, part of the Johnson & Johnson Medical Devices Companies has announced post-approval procedures were successfully performed with the Heliostar radiofrequency balloon ablation catheter at sites across Europe. In Europe, the Heliostar Balloon Catheter is indicated for use...
Benjamin D’Souza, assistant professor of Medicine at the University of Pennsylvania, cardiac electrophysiologist at Penn Presbyterian Medical Center, Philadelphia, USA, discusses the recent developments in robotics in the electrophysiology (EP) lab.
The two main criticisms of the use of robotics...
The Heart Rhythm Society (HRS) has announced that its cardiovascular digital health conference—HRX—has been rescheduled to September 2022. The event had been due to take place in San Diego, USA, over 2–4 December 2021.
In a statement issued late last...
Specific and dynamic changes on electrocardiograms (ECGs) of hospitalised patients with COVID-19 or influenza can help predict a timeframe for worsening health and death, according to a new study published in the American Journal of Cardiology. The study, carried...
Continuous heart rhythm monitoring—with anticoagulation if atrial fibrillation (AF) is detected—does not prevent strokes in those at risk. That is the finding of the LOOP study, presented during a Hot Line session at the European Society of Cardiology’s 2021...
Ablation plus cardiac resynchronisation therapy (CRT) is superior to pharmacological rate control in reducing mortality in severely symptomatic permanent atrial fibrillation (AF) patients with a narrow QRS, according to late breaking research presented in a Hot Line session at...
Image-guided fibrosis ablation in addition to pulmonary vein isolation (PVI) does not improve ablation success rates compared to PVI alone in patients with persistent atrial fibrillation (AF), according to an intention-to-treat analysis presented in a Hot Line session at...
The Arrhythmia Alliance’s 2021 Heart Rhythm Congress (A-A HRC 2021 3–6 October, virtual) returns for its second ever digital edition, which will see more than 250 world heart rhythm specialists deliver more than 200 hours of educational content.
In recognition...
Galaxy Medical announced today the initiation of the SPACE-AF study with enrolment of the first two patients at Southlake Regional Health Centre in Newmarket, Canada.
In the study, the Centauri pulsed electric field (PEF) ablation system will be used to...
QardioDirect, the all-inclusive service for remote patient monitoring (RPM) from Qardio, now includes ambulatory cardiac monitoring to help diagnose atrial fibrillation (AF) along with a range of other arrhythmias.
Multiple short duration recordings can identify symptomatic and asymptomatic periods of...
VitalConnect has announced the launch of a new version of VistaCenter for cardiac and remote patient monitoring. This new version brings updates including simplified patient intake, live patient census views, custom notification profiles and report delivery—all designed to streamline...
Medtronic has announced a pilot programme with Mpirik to address disparities in care associated with the prevention of sudden cardiac arrest (SCA), a condition in which the heart suddenly and unexpectedly stops beating. SCA is caused by a disturbance...
Zoll Medical and Itamar Medical have announced that the two companies signed a definitive agreement under which Zoll Medical will acquire all outstanding ordinary shares of Itamar Medical for a total value of approximately US$538 million.
Itamar Medical is focused...
Biotronik has announced the first observational results of the CERTITUDE registry, documenting real-world clinical outcomes of patients in the USA who use implantable loop recorders (ILR), pacemakers, implantable cardioverter-defibrillators (ICDs), and cardiac resynchronisation therapy (CRT-D) devices that have Biotronik...
Stereotaxis and Shanghai Microport EP Medtech have announced a broad collaboration to advance technology innovation and commercial adoption of robotics in electrophysiology in China.
The agreement brings together MicroPort EP’s commercial and product leadership in China’s electrophysiology market with Stereotaxis’...
Bhargav Vemulapalli, Shreya Srivastava and John Kassotis (New Brunswick, USA) review the potential use of antibiotic-eluting envelopes (ABEs) based on available clinical data, while underscoring the need for standardised guidelines related to antibiotic prophylaxis.
Infections associated with cardiac implantable electronic...
AliveCor has announced the results of what it describes as the largest study to assess the reliability of the detection of atrial fibrillation (AF) through commercially available personal ECG devices, in which its KardiaMobile 6L device was found to...
Cardiomatics, the cloud AI tool that allows any trained medical staff to carry out electrocardiogram (ECG) analysis, has raised US$3.2 million in seed funding led by KAYA, the Central and Eastern European VC which has backed companies including Rohlik,...
Edoxaban is noninferior to warfarin and its analogues for adverse clinical events in patients with atrial fibrillation after transcatheter aortic valve implantation (TAVI), new research has found.
Findings of the ENVISAGE-TAVI AF study, which were published in the New England...
Medtronic has announced new data from the Micra Coverage with Evidence Development (CED) study, which showed the Micra transcatheter pacing system (TPS) was associated with a 38% reduction in reinterventions and a 31% reduction in chronic complications at two...
Biosense Webster, part of the Johnson & Johnson Medical Devices Companies, has announced the presentation of data from the inspIRE clinical trial at the European Society of Cardiology’s 2021 congress (ESC 2021, 27–30 August, virtual).
inspIRE is a prospective, multicentre,...
The European Society of Cardiology (ESC) Guidelines on cardiac pacing and cardiac resynchronisation therapy (CRT) have been published online in European Heart Journal.
The use of pacemakers continues to grow as populations worldwide are living longer. It is estimated that...
Abbott has announced late-breaking data from the Amulet LAA Occluder IDE trial, a multicentre, head-to-head study comparing the company's Amplatzer Amulet left atrial appendage (LAA) occluder with the Watchman (Boston Scientific) device to treat patients with atrial fibrillation (AF)...
Abbott has announced results of the landmark GUIDE-HF clinical trial, a 1,000 patient randomised study designed to assess the benefits of the CardioMEMS HF System in people living with NYHA Class II, III and IV heart failure.
Data adjusted for...
The world’s first feasibility study has found that drones can be used to deliver defibrillators to people with suspected cardiac arrest in the community. The research is presented at the European Society of Cardiology’s 2021 congress (ESC 2021, 27–30...
The European Society of Cardiology (ESC) Guidelines for the diagnosis and treatment of acute and chronic heart failure are published online today in European Heart Journal. This was the first ESC Guideline to include patients as full members of...
A six-month exercise programme helps maintain normal heart rhythm and reduces the severity of symptoms in patients with atrial fibrillation (AF), according to late-breaking research to presented this week at the European Society of Cardiology’s 2021 congress (ESC 2021,...
Ultromics, a developer of AI enabled cardiovascular imaging solutions, has announced that it has raised US$33 million in a Series B funding round. The round was led by the Blue Venture Fund with participation from Optum Ventures, GV, and...
The US Food and Drug Administration (FDA) has granted breakthrough device designation to Abiomed’s Impella ECP expandable percutaneous heart pump. The designation means the FDA will prioritise Impella ECP’s regulatory review processes including design iterations, clinical study protocols and...
CardioNXT has received marketing clearance from the US Food and Drug Administration (FDA) for the platform technology comprised of the iMap 3D Navigation & Mapping system, Activate software, Sensor Enabled Axis Patient Patches, and MultiLink sensor enabled catheter.
The CardioNXT...
Biotronik has announced the results from a new study published in EP Europace this week confirming that heart failure (HF) decompensation can be predicted early when monitored using an algorithm that combines existing remote monitoring trends and baseline risk...
People who work night shifts are at increased risk of developing atrial fibrillation (AF), according to research published in the European Heart Journal.
The study is the first to investigate the links between night shift work and AF, and uses...
Abbott has announced that the US Food and Drug Administration (FDA) has approved the company's Amplatzer Amulet Left Atrial Appendage Occluder to treat people with atrial fibrillation (AF) who are at risk of ischaemic stroke. The device offers immediate...
Acutus Medical has received US Food and Drug Administration (FDA) and CE mark clearance for its suite of software upgrades known collectively as AcQMap 8. The upgrades introduce new mapping algorithms into Acutus’ foundational technology, the AcQMap 3D imaging...
Long-term continuous electrocardiogtraphy (LT-ECG), coupled with oversight by a physician, significantly outperforms algorithm-dependent mobile cardiac telemetry (MCT) in the detection of significant arrhythmias according to the findings of a study presented during a late-breaking trial session at the 2021...
Caption Health has announced that the Centers for Medicare and Medicaid Services (CMS) have approved new technology add-on payments (NTAP) for the Caption Guidance AI-based software platform for Medicare patients receiving in-patient care.
Cases eligible for NTAP will have a...
Women remain underrepresented in cardiovascular clinical trials despite initiatives to ensure broader inclusivity. This is according to two reports, both published in the last week, suggesting that lack of representation may limit availability of treatment data for combating cardiovascular...
The challenge of rapid detection and diagnosis of atrial fibrillation (AF) has been made even harder with the onset of the COVID-19 pandemic, and the resulting disruption to appointments and restrictions of access to healthcare services. Against this backdrop,...
Economic analyses from the WRAP-IT study, sponsored by Medtronic, demonstrate the Tyrx cardiac absorbable antibacterial envelope’s (Tyrx Envelope) cost effectiveness in European markets for patients at increased risk of infections.
This follows the publication of a study in Heart Rhythm...
Rhythm control therapy offers a clinical benefit when initiated within one year of a diagnosis of atrial fibrillation (AF) in patients with signs or symptoms of heart failure, a sub-analysis of the EAST–AFNET 4 trial has concluded.
The findings were...
A new analysis of the CABANA (Catheter ablation versus antiarrhythmic drug therapy for atrial fibrillation) suggests that catheter ablation offers positive economic outcomes compared to drug therapy for the treatment of atrial fibrillation (AF). This is according to the...
Results of the PAUSE-SCD trial show a significant reduction in risk of ventricular tachycardia (VT) recurrence, cardiovascular hospitalisation, and death when catheter ablation is used as an early first-line treatment for patients with structural heart disease.
The PAUSE-SCD trial is...
Use of direct oral anticoagulants (DOACs) in patients undergoing ventricular tachycardia radio frequency ablation (RFA) is associated with a reduced risk of transient ischaemic attack or stroke, and asymptomatic, magnetic resonance imaging (MRI) detected cerebrovascular event.
This is according to...
Abbott has announced the US launch of Jot Dx, the company’s latest insertable cardiac monitor (ICM). In a press release, Abbott said that the Jot Dx ICM gives clinicians and hospitals control of how they manage the flow of...
Medtronic has announced US Food and Drug Administration (FDA) clearance for two AccuRhythm AI algorithms for use with the LINQ II insertable cardiac monitor (ICM). The algorithms address the two most common ICM false alerts—atrial fibrillation (AF) and asystole.
AccuRhythm...
AliveCor and Acutus Medical will collaborate to assess the integration of data collection tools across the cardiology continuum of care, which the companies say may help guide decision making in the management of cardiac arrhythmia patients.
The collaboration includes a...
Angel Medical Systems (AngelMed) has announced the first commercial implantation and US launch of its The Guardian device. The procedure marks the Guardian's first use following its recent US Food and Drug Administration (FDA) approval. Indicated for acute coronary...
Affera has announced the first-in-human use of its circumferential pulmonary vein isolation (PVI) pulsed field ablation (PFA) catheter, SpherePVI.
The work was conducted as part of a multicentre first-in-human study designed to evaluate the safety and effectiveness of the SpherePVI...
MicroPort Cardiac Rhythm Management (CRM) has entered into definitive agreements in connection with its Series C financing with total investment proceeds of US$150 million.
Hillhouse Capital Group and MicroPort will co-lead the Series C investment and will invest US$20 million...
Medtronic has announced new data from the WRAP-IT study published in Heart Rhythm, which it said demonstrate a significantly lower infection risk for patients who develop haematomas after cardiac implantable electronic devices (CIEDs) when the Tyrx absorbable antibacterial envelope...
People who made even small increases in their daily physical activity levels after receiving an implantable cardioverter defibrillator (ICD) experienced fewer incidences of hospitalisation and had a decreased risk of death, according to research published in Circulation: Cardiovascular Quality...
There is no evidence that moderate coffee consumption can cause cardiac arrhythmia, according to the authors of a prospective, population-based community cohort study, findings of which were published this week in JAMA Internal Medicine.
According to authors Eun-jeong Kim (University...
Two-year results from the PINNACLE FLX clinical trial assessing the safety and efficacy of the next-generation Watchman FLX (Boston Scientific) left atrial appendage closure (LAAC) device for patients with non-valvular atrial fibrillation (NVAF) have shown that patients implanted with...
Ancora Heart has closed US$80 million in equity financing, with the funds to be used to accelerate the CORCINCH-HF pivotal clinical study of the AccuCinch ventricular restoration system. The device is designed to treat patients who have symptomatic heart...
The authors of research published in JACC: Clinical Electrophysiology claim that their findings will help physicians to identify, treat and prevent atrial fibrillation (AF) in patients with hypertrophic cardiomyopathy (HCM).
The research, led by Christopher Kramer the chief of the...
atHeart Medical has announced the successful treatment of the first five patients in its ASCENT ASD US Investigational Device Exemption (IDE) pivotal trial. This study, the company's first in the USA, will evaluate the safety and efficacy of the...
AliveCor has today announced the receipt of 510(k) clearance from the US Food and Drug Administration (FDA) for healthcare professionals to use the KardiaMobile 6L device to calculate patients' QTc interval. This FDA action follows the enforcement policy for...
East End Medical has announced it has received US Food and Drug Administration (FDA) clearance for the company's SafeCross transseptal radiofrequency (RF) puncture & steerable balloon introducer system. The three-in-one system, which includes a steerable introducer sheath with an...
Among racial or ethnic minority patients enrolled in the North American cohort of the CABANA (Catheter ablation versus antiarrhythmic drug therapy for atrial fibrillation) trial, catheter ablation significantly improved major clinical outcomes compared with drug therapy. This is according...
Fred Kusumoto (Jacksonville, USA), current president of the Heart Rhythm Society (HRS), sits down with Cardiac Rhythm News to discuss the upcoming Heart Rhythm 2021 meeting which, for the first time, will be a hybrid event consisting of both...
Adagio Medical has announced successful first-in-human use of ultra-low temperature cryoablation (ULTC) for the treatment of monomorphic ventricular tachycardia (VT).
The two-hour mapping and ablation procedure was performed by Tom De Potter, associate director, Cardiovascular Center Department of Cardiology, Electrophysiology...
Royal Philips has announced the first procedure using its real-time 3D intracardiac echocardiography (ICE) catheter. The left atrial appendage occlusion (LAAO) procedure was carried out by Mohamad Adnan Alkhouli (Mayo Clinic, Minnesota, USA).
Used with Philips Premium Cardiology Ultrasound...
The risk of heart damage from COVID-19 infection is far greater than that from receiving a dose of the messenger RNA (mRNA) vaccine, according to the authors of a retrospective case series published in JAMA Cardiology.
Researchers from Mayo Clinic...
Boston Scientific has exercised its option to acquire the remaining shares of Farapulse. The acquisition will complement the existing Boston Scientific electrophysiology portfolio to include the Farapulse Pulsed Field Ablation (PFA) system—a non-thermal ablation system for the treatment of...
Medtronic has announced the initiation of DEFINE AFib, an app-based research study which aims to address unanswered questions around atrial fibrillation (AF) burden and its impact on patient outcomes, quality of life, and healthcare utilisation. The study will use...
Angel Medical Systems (AngelMed) has announced US Food and Drug Administration (FDA) approval of the second-generation AngelMed Guardian device. The AngelMed Guardian system is an implantable cardiac detection monitor and patient-warning system for acute coronary syndrome (ACS) events, including...
The American Heart Association (AHA) has this week issued a scientific statement on the management of atrial fibrillation (AF) in patients with heart failure and reduced ejection fraction (HFrEF).
Published in Circulation: Arrhythmia and Electrophysiology, the statement reviews available evidence...
CardioFocus has announced that the Japanese Ministry of Health, Labor and Welfare (MHLW) has approved the company's HeartLight X3 System for marketing in Japan. The HeartLight X3 System is CardioFocus' catheter ablation technology for controlled and consistent pulmonary vein...
Medtronic has received US Food and Drug Administration (FDA) expanded approval for the Arctic Front family of cardiac cryoablation catheters for the treatment of recurrent symptomatic paroxysmal atrial fibrillation (AF) as an alternative to antiarrhythmic drug (AAD) therapy as...
The Heart Rhythm Society (HRS) has announced the appointment of Simintha Esson as its new chief strategy officer (CSO).
In this role, Esson is responsible for developing and implementing strategies to support the organisation’s short- and long-term business development initiatives....
The president of the European Society of Cardiology (ESC) has called for greater awareness of the signs of cardiac arrest and how to respond, following the collapse of Danish footballer Christian Eriksen, during a European Championship match this week.
Eriksen,...
Imperial College Healthcare NHS Trust (London, UK) is working with remote healthcare developer, Luscii, to provide an at-home monitoring platform to support patients with heart failure.
In its initial phase, the Luscii platform has been rolled out to a distinct...
MicroPort CRM has announced the European launch of Alizea and Borea pacemakers after receiving the CE mark under the new Medical Device Regulation. The devices are equipped with Bluetooth technology for streamlined remote monitoring when paired with the SmartView...
Robert Kowal is the chief medical officer of the Cardiac Rhythm businesses at Medtronic. In this interview he shares his views and experiences on the role that remote monitoring has taken in patients’ lives, both in the past and...
A campaign by Arrhythmia Alliance to DETECT atrial fibrillation (AF)—the most common type of arrhythmia—found that 6% of people aged 65 or older attending a COVID-19 vaccination centre had “possible AF”. The result supports previous findings that the incidence...
Among patients with both heart failure and atrial fibrillation (AF), treatment strategies focused on rhythm control, using catheter ablation, and those focused on rate control, using drugs and or a pacemaker, showed no significant differences in terms of death...
A new physiological measurement of heart function could improve survival for people with heart failure by identifying high-risk patients who require tailored treatments, a study published in the journal Heart, Lung and Circulation suggests.
The study is the first to...
GE Healthcare is collaborating with the American College of Cardiology (ACC) through support of and participation in the ACC’s Applied Health Innovation Consortium for the purpose of building a roadmap for Artificial Intelligence (AI) and digital technology in cardiology...
Medtronic has announced clinical trial results from the STROKE AF trial demonstrating the superiority of the Reveal LINQ insertable cardiac monitor (ICM) to detect abnormal heartbeats, otherwise known as atrial fibrillation (AF), in both large and small vessel stroke...
Abbott has announced it has received CE mark and Health Canada approval for its Amplatzer steerable delivery sheath, which is used with the company's Amplatzer Amulet left atrial appendage (LAA) occluder. Now available in Europe and Canada, the device...
Medtronic is stopping the distribution and sale of the Medtronic HVAD system. This morning, the company notified physicians to cease new implants of the HVAD System and transition to an alternative means of durable mechanical circulatory support.
Medtronic also announced...
Siemens Healthineers has announced that it has received CE Mark for the AcuNav Volume ICE (intracardiac echocardiography) catheter, an imaging guide that provides real-time, wide-angle visualisation of heart anatomy during structural heart and electrophysiology procedures.
“The AcuNav Volume ICE catheter...
Abiomed has acquired preCARDIA, developer of a proprietary catheter and controller to expand options for patients with acute decompensated heart failure (ADHF). The preCARDIA system is designed to rapidly treat ADHF-related volume overload by reducing cardiac filling pressures and...
Implicity has announced a distribution agreement with MicroPort CRM, through which MicroPort CRM can sell and distribute Implicity's cloud-based solution in the USA as part of its remote patient monitoring offerings with a new digital service that allows cardiologists...
Physical activity that conforms to medical and health association guidelines is associated with a lower risk of atrial fibrillation (AF) and stroke, according to a study by analysing the activity of nearly 100,000 individuals equipped with wrist-worn accelerometers to...
The European Union (EU) Medical Devices Regulation (MDR) takes effect from today (26 May 2021).
The Regulation revises quality and safety standards and the range of regulated devices and was first initiated in May 2017, with an initial three-year transition...
iRhythm Technologies has announced two US Food and Drug Administration (FDA) 510K clearances—one for a new and improved design of its flagship monitor and a second for updated artificial intelligence (AI) capabilities.
The new Zio monitor is designed to significantly...
Cardiomatics and the Medical University of Warsaw, Warsaw, Poland are developing a tool for automatic assessment, analysis, and interpretation of electrocardiographic signals (ECG) from paediatric patients.
The Cardiomatics Junior Project is a direct response to the identified lack of ECG...
The US Food and Drug Administration (FDA) has issued advice to patients with implanted devices such as pacemakers or implantable defibrillators to take precautions against the risk of magnetic interference from smartphones and smart watches.
According to the FDA, some...
The proportion of cardiologists reporting burnout has nearly doubled during the COVID-19 pandemic, according to research presented at the American College of Cardiology’s 70th Annual Scientific Session (ACC.21, 15–17 May, virtual). This was among the key findings of the...
Transcatheter left atrial appendage occlusion (LAAO) with a Watchman (Boston Scientific) device was associated with a low rate of stroke at one year, even among older patients with atrial fibrillation (AF) who faced a high risk for stroke or...
Patients with an elevated risk of stroke due to arrhythmia or atrial fibrillation (AF), were much less likely to suffer a stroke after undergoing heart surgery if doctors concurrently performed left atrial appendage occlusion (LAAO), according to the results...
An electrocardiogram (EKG)-based artificial intelligence (AI) algorithm based can enable the early diagnosis of low ejection fraction in patients in routine primary care, according to research published in Nature Medicine.
The ECG AI-Guided Screening for Low Ejection Fraction—EAGLE—trial set out...
Pulmonary vein isolation (PVI) with a “single-shot” pulsed-field ablation (PFA) catheter results in excellent PVI durability and acceptable safety with a low one-year rate of atrial arrhythmia recurrence in paroxysmal atrial fibrillation (AF) patients, according to data pooled from...
Acutus Medical has announced CE mark approval for a broad suite of electrophysiology (EP) products including the AcQCross family of universal transseptal crossing devices, the next generation AcQGuide MAX and VUE large bore delivery sheaths and the next generation...
Discrimination and harassment are a common experience among those working in cardiology, according to the findings from a global survey carried out by the American College of Cardiology (ACC), which have been published in the Journal of the American...
Galaxy Medical (Galaxy) and Japan Lifeline (JLL) have announced an exclusive distribution agreement for the ALPHA1 ablation catheter in the USA, developed by Japan Lifeline for use with the Galaxy Centauri pulsed electric field (PEF) system. Under the terms...
A significant proportion of cardiology trainees are uncomfortable with using telemedicine and feel that better preparation for new-tech medicine is needed, findings of a survey published in the Canadian Journal of Cardiology indicate.
The use of telemedicine has become routine...
Abbott has announced a new trial focused on improving the treatment for people simultaneously battling both atrial fibrillation (AF) and heart failure.
The TAP-CHF trial (Evaluating the Treatment of Atrial Fibrillation in Preserved Cardiac Function Heart Failure) aims to discover...
Subcutaneous implantable cardioverter defibrillator (S-ICD) devices have demonstrated a cardioversion efficacy rate of 98% over a period of at least five years in the EFFORTLESS study—the results of which were presented in a late-breaking presentation at the 2021 European...
AtriCure has announced US Food and Drug Administration (FDA) approval of the EPi-Sense system to treat patients diagnosed with long-standing persistent atrial fibrillation (AF).
The CONVERGE trial demonstrated superiority in the hybrid AF therapy arm compared to endocardial catheter ablation...
Patients hospitalised with COVID-19 may be at risk of developing heart failure even if they do not have a previous history of heart disease or cardiovascular risk factors, a study published in the Journal of the American College of...
Further efforts should be made to increase participation in atrial fibrillation (AF) screening—particularly in elderly populations, where systematic screening for AF can help to lower the risk of adverse events and reduce comorbidities. This is according to a late-breaking...
Patients receiving an implantable cardioverter defibrillator (ICD) should be regularly screened for anxiety and depression, according to research presented at the European Heart Rhythm Association annual meeting (EHRA 2021, 23–25 April, online).
Study author Susanne Pedersen of Odense University Hospital,...
A smartphone-based electrocardiogram (ECG) screening accurately detected previously unknown atrial fibrillation (AF) in American Indians, and more than half who were diagnosed were younger than the recommended screening age of 65, according to new research published in the Journal...
Atrial fibrillation (AF) could be detected during annual foot assessments in patients with diabetes, according to research presented today at the European Heart Rhythm Association annual meeting (EHRA 2021, 23–25 April, online).
The finding was presented by Ilias Kanellos of...
This article was sponsored by Biosense Webster
Putting the patient at the centre of the discussion is vital to setting the right strategy for the treatment of atrial fibrillation (AF). This is according to Biosense Webster’s recently published report: Atrial...
A project at Oxford University Hospitals NHS Foundation Trust (Oxford, UK) to test how effective smart devices are at detecting heart rhythm problems has received a grant of almost £150,000 from national charity Heart Research UK.
Atrial fibrillation (AF) is...
Acutus Medical has announced US Food and Drug Administration (FDA) clearance of the AcQCross family of universal transseptal crossing devices. The transseptal puncture system is specifically engineered to pair with Acutus’ own suite of sheaths and with sheaths sold...
Cardialen has announced the publication of a first-in-human study in the Journal of the American College of Cardiology: Clinical Electrophysiology investigating its low-voltage MultiPulse therapy to terminate atrial fibrillation (AF). The study consisted of 42 patients who were indicated...
The charity Arrhythmia Alliance has launched the Arrhythmia Alliance Healthcare Pioneers Report—Showcasing Best Practice in SVT to provide examples of good care to improve outcomes and quality of life of people with supraventricular tachycardia (SVT).
SVT is a rapid increase...
More patients, particularly those with medical risk factors or from underserved communities, opted into telehealth appointments for their cardiovascular care during the COVID-19 pandemic, a cross-sectional study of more than 176,000 ambulatory cardiology visits in Los Angeles County, USA,...
AF Association has launched an opportunistic screening programme to DETECT atrial fibrillation (AF) at COVID-19 vaccination clinics across the UK. The charity has created an online resource hub for healthcare professionals working at vaccine clinics to use to detect...
The American College of Cardiology (ACC) has launched the EP Device Implant Registry, which will include data on ICD and CRT-D procedures previously captured in the NCDR ICD Registry as well as provide the flexibility to capture novel pacemaker...
Acutus Medical has announced initial US enrolment in the company’s AcQForce Flutter Investigational Device Exemption (IDE) clinical trial. The trial is expected to enrol up to 150 subjects in centres globally and will evaluate the safety and efficacy of...
Dipti Itchhaporia has today begun her term as president of the American College of Cardiology (ACC). During her one-year presidency, she will lead the over 54,000-member global organization in its mission to transform cardiovascular care and improve heart health.
Itchhaporia...
Patients with valvular atrial fibrillation (AF) who were new users of direct oral anticoagulants (DOACs) were at lower risk for ischaemic stroke or systemic embolism and major bleeding than new users of warfarin, an analysis published in the Annals...
More than a third of female cardiologists polled in a UK survey on discrimination and sexism in the profession have reported receiving unwanted sexual comments, attention or advances from a colleague. The findings of the survey were published online...
Tempus has announced that the US Food & Drug Administration (FDA) has granted the company Breakthrough Device Designation for its ECG analysis platform. The platform, developed in collaboration with Geisinger, aids clinicians in identifying patients at increased risk of...
Daiichi Sankyo has announced that it has entered into a collaborative agreement with three technology partners—FibriCheck, neoHealthTech and Capitol Medicare—to improve timely detection and diagnosis of atrial fibrillation (AF).
The partners will be working in collaboration to offer digital solutions...
Bristol Myers Squibb has announced that the US Food and Drug Administration (FDA) has accepted its new drug application (NDA) for mavacamten, an investigational, novel, oral, allosteric modulator of cardiac myosin, for patients with symptomatic obstructive hypertrophic cardiomyopathy (oHCM)....
Higher procedural volume is associated with better outcomes for percutaneous left atrial appendage occlusion (LAAO) procedures, research published online in JACC: Cardiovascular Interventions has found.
Outlining the rationale for the study, lead author Salik Nazir (University of Toledo Medical Center,...
An assessment method known as the index-beat approach could improve the diagnosis of heart failure in patients with atrial fibrillation (AF), authors of a study published online in BMJ: Heart have found.
Karina Bunting (Institute of Cardiovascular Sciences, University of...
Medtronic has announced CE mark approval for the SelectSite C304-HIS deflectable catheter system, as well as CE mark labelling expansion for the SelectSecure MRI SureScan Model 3830 cardiac pacing lead for use in His-bundle pacing procedures.
Permanent His-bundle pacing is...
Far from being the first event in the electrophysiology calendar now to make the switch to a digital-only format, the 2021 edition of the Atrial Fibrillation Symposium (AFIB2021, 18 March, virtual) will be the latest to make its digital...
Qardio has announced that the US Food and Drug Administration (FDA) has granted the company 510(k) clearance for its QardioCore ambulatory ECG device. QardioCore will initially be marketed for holter monitoring applications, for use with QardioMD, Qardio's cloud-based remote...
Five-year follow-up results from the Gore REDUCE study, assessing the Gore Cardioform or Helex septal occluder versus antiplatelet therapy in long-term recurrent stroke prevention, have been published in the March 2021 issue of The New England Journal of Medicine (NEJM). The results...
The American College of Cardiology (ACC) has announced a number of the late-breaking clinical trials to be presented at its 70th annual scientific session (ACC.21, 15–17 May, virtual).
The event has recently been switched to an all-virtual format having originally...
CorSystem has announced the first use of its Bipolar Ablation Adapter since the device received CE mark in July 2020.
The patient who underwent treatment was suffering from a VT storm which required frequent interventions of implantable cardioverter-defibrillator. The patient...
Inflammatory heart disease is a rare finding among professional athletes with mild or asymptomatic COVID-19 infection, a large-scale study has found. The study, led by Columbia University Vagelos College of Physicians and Surgeons (New York, USA) in collaboration with...
ControlRad has announced US Food and Drug Administration (FDA) 510(k) clearance to market ControlRad Select, a technology that utilises proprietary semi-transparent filters, a user-interface tablet, and image processing algorithms to reduce radiation exposure during fluoroscopically guided procedures.
The technology is...
Medtronic has announced the first procedures in the investigational device exemption (IDE) pivotal trial to evaluate the PulseSelect pulsed field ablation (PFA) system, a novel, breakthrough technology that uses pulsed electric fields to treat atrial fibrillation (AF).
The first procedure...
Atrial fibrillation (AF) burden at six months after catheter ablation is predictive of all-cause mortality and heart failure hospitalisation, in AF patients with heart failure and low ejection fraction, according to the findings of a subanalysis of the CASTLE-AF...
A global collaboration between international heart failure bodies seeks to standardise the language and practices around the definition and classification of heart failure around the globe.
The Heart Failure Society of America (HFSA), the Heart Failure Association of the European...
An open-source platform, OpenEP, has been made available to advance research on atrial fibrillation (AF), which developers claim can perform 90% of the analyses performed in contemporary electrophysiology studies. The platform, which is detailed in a study recently published...
FARAPULSE has announced that the first patients have been treated in the ADVENT trial, a US Food and Drug Administration (FDA) Investigational Device Exemption (IDE) pivotal trial to evaluate the safety and effectiveness of its pulsed field ablation (PFA)...
iRhythm Technologies has announced the results of the SCREEN-AF study, led by researchers at Sunnybrook Health Sciences Centre in Toronto, Canada and University Hospital in Leipzig, Germany, published in JAMA Cardiology.
The transatlantic clinical trial found that the use of...
Medtronic has announced new results from the REVERSE trial, evaluating outcomes of cardiac resynchronisation therapy (CRT) for patients with mild heart failure (HF). The analysis of the REVERSE (REsynchronization reVErses Remodeling in Systolic left vEntricular dysfunction) clinical trial shows...
CardioFocus has announced that results from the HeartLight X3 Pivotal Study have been published online in the journal Circulation: Arrhythmia and Electrophysiology (CircEP), showing that all endpoints of the pivotal study were achieved.
The results demonstrated significantly faster pulmonary vein...
Piccolo Medical has announced that it has received US Food and Drug Administration (FDA) clearance of its SmartPICC system.
The SmartPICC system offers the ability to navigate peripherally inserted central catheters (PICCs) to the entryway of the heart without the...
The American College of Cardiology (ACC) has announced that its 70th annual scientific session, will be held as an all virtual meeting. The meeting had been due to be held as a hybrid event, across 15–17 May, with in-person...
Urgent action is needed to address the growing burden of heart failure and the impact of COVID-19 in the UK, according to a report issued this week by the Alliance for Heart Failure.
The report—‘Heart Failure: A call to action’—makes...
RhythMedix has announced the release of its next-generation RhythmStar wearable device with built-in 4G cellular connectivity. The novel cardiac telemetry monitor is worn for extended remote monitoring without the need for an additional phone or communication device.
The technology enables...
MicroPort CRM has launched an online education website—MicroPort Academy CRM—dedicated to cardiac pacing therapy and the functionality of MicroPort CRM products.
MicroPort Academy CRM provides personalised and interactive training plans, ranging from diagnosis of ECG tracings to techniques of pacemaker...
The number of patients with heart failure worldwide nearly doubled from 33.5 million in 1990 to 64.3 million in 2017 according to research published in the European Journal of Preventive Cardiology.
The analysis used data from the Global Burden of...
Updated recommendations on the optimal antithrombotic treatment of patients with atrial fibrillation (AF) receiving oral anticoagulants after undergoing percutaneous coronary intervention (PCI) have been published this week in the journal Circulation.
The recommendations are the latest in a series of...
MedLumics has announced that it has closed an €18 million (US$21.7 million) financing round and has appointed Rich Ferrari as Chairman of the Board of Directors.
The company is developing AblaView, an optically-guided real-time ablation catheter system for the treatment...
CardiacSense has received the CE mark to market and sell a medical grade watch which can detect and remotely monitor atrial fibrillation (AF) and heart rate variability (HRV). The indications certified include detection of AF and monitoring of HRV...
Royal Philips has announced that it has completed the acquisition of BioTelemetry, a US provider of remote cardiac diagnostics and monitoring.
The acquisition of BioTelemetry is a strong fit with Philips’ cardiac care portfolio, and its strategy to transform the...
A study published in the European Journal of Preventive Cardiology provides “strong evidence” of a causal relationship between elevated blood pressure and atrial fibrillation (AF). Authors of the study suggest that strict blood pressure control measures could be an...
B-Secur has announced that it has received US Food and Drug Administration (FDA) 510(K) clearance for its HeartKey software library.
B-Secur’s HeartKey is a suite of EKG/ECG algorithms that combine user identification, health and wellness to generate accurate data encrypted...
The merits of catheter ablation as a first-line treatment for atrial fibrillation (AF) were considered during the second day of AF Symposium 2021 (29–31 January, virtual), which brought together key data from recent clinical trials assessing ablative approaches to...
Hybrid ablation, a procedure which involves “attacking” the atrial substrate from the epicardium and endocardium, has been shown to result in significantly higher freedom from arrhythmia compared to conventional catheter ablation, in persistent and long-standing persistent atrial fibrillation (AF)—late-breaking...
Findings of two studies investigating the use of a lattice-tip catheter for the treatment of atrial fibrillation (AF) were presented during a late-breaking trial session at AF Symposium (29–31 January, virtual) by Vivek Reddy (Icahn School of Medicine at...
Researchers using artificial intelligence (AI) have determined that a smartphone-enabled mobile EKG device can accurately determine a patient’s QT. This can be used to identify patients at risk of sudden cardiac death from congenital long QT syndrome (LQTS) or...
Results of a sub-analysis of the CONVERGE clinical trial, presented during a late-breaking trial session at the digital AF Symposium (29–31 January, virtual), suggest that the hybrid Convergent procedure yields superior outcomes to catheter ablation in patients with long-standing...
Medtronic has received US Food and Drug Administration (FDA) approval for the DiamondTemp ablation (DTA) system which treats patients with recurrent, symptomatic paroxysmal atrial fibrillation (AF) and who have been unresponsive to drug therapy. The DiamondTemp system is the...
Galaxy Medical, a developer of pulsed electric field (PEF) technology for the treatment of cardiac arrhythmias, has announced that the proprietary Centauri system will be featured in a recorded case presentation during the AF Symposium (29–31 January, virtual).
Ante Anić,...
CathVision ApS has announced that its novel cardiac electrophysiology system and artificial intelligence platform to guide ablation therapy for heart rhythm disorders, will be featured at the 26th annual international AF Symposium (29–31 January, virtual).
Josef Kautzner, Institute of Cardiology...
Acutus Medical has announced a number presentations relating to its mapping and ablation procedures during 26th Annual International AF Symposium (29–31 January, virtual). The studies demonstrate the beneficial capabilities of guided ablation therapy utilising the company’s entire diagnostic and...
Farapulse announced that it has received the CE mark for its pulsed field ablation (PFA) system for the treatment of paroxysmal atrial fibrillation (AF). The approval will allow the company to commercialise the cardiac PFA system and permits marketing...
RHYTHM AI has announced publication of a clinical outcome study for its STAR (Stochastic Trajectory Analysis of Ranked signals) Mapping system, which is designed to improve outcomes in patients receiving ablation treatment for persistent atrial fibrillation (AF). The article...
Cardialen has been awarded a US$2.8 million National Institutes of Health (NIH) phase II SBIR grant to further the clinical study of low-energy cardioversion of atrial fibrillation (AF) in human subjects in the USA.
The grant was awarded after a...
Kardium has raised US$115 million in a new financing round, which will be used to accelerate commercial growth of the Globe mapping and ablation system for the treatment of atrial fibrillation (AF) across Europe and to conduct a clinical...
Following the announcement of CE mark approval for the Aktiia cuffless blood pressure monitor bracelet, Gregoire Wuerzner (Lausanne University Hospital and University of Lausanne, Switzerland) clinical advisor to Aktiia and President of the Swiss Society of Hypertension, discusses the...
The wealthiest countries in Europe have higher death rates from atrial fibrillation (AF) than the least wealthy and these death rates are increasing more rapidly than incidence rates, according to an analysis published in the European Heart Journal. The...
Aktiia has announced that its automated blood pressure monitoring system has received the CE mark as a Class IIa medical device. With the CE Mark, Aktiia now has access to over 40 countries worldwide.
Aktiia says that the device is...
InCarda Therapeutics has announced dosing of the first US patient in the company’s multinational INSTANT Phase 2 clinical trial of InRhythm (flecainide for inhalation) in patients with recent-onset paroxysmal atrial fibrillation (PAF). The trial is currently being conducted at...